
Cialis ist bekannt für seine lange Wirkdauer von bis zu 36 Stunden. Dadurch unterscheidet es sich deutlich von Viagra. Viele Schweizer vergleichen daher Preise und schauen nach Angeboten unter dem Begriff cialis generika schweiz, da Generika erschwinglicher sind.
Jn 7(2).pdf
of Obsessive Compulsive Disorder
D. Corydon Hammond, PhD
ABSTRACT. Introduction. Blinded, placebo-controlled research (e.g.,
Sterman, 2000) has documented the ability of brainwave biofeedback to
recondition brain wave patterns. Neurofeedback has been used success-
fully with uncontrolled epilepsy, ADD/ADHD, learning disabilities,
anxiety, and head injuries. However, nothing has been published on the
treatment of obsessive-compulsive disorder (OCD) with neurofeedback.
Method. Quantitative EEGs were gathered on two consecutive OCD
patients who sought treatment. This assessment guided protocol selec-tion for subsequent neurofeedback training.
Results. Scores on the Yale-Brown Obsessive-Compulsive Scale and
the Padua Inventory normalized following treatment. An MMPI was ad-ministered pre-post to one patient, and she showed dramatic improve-ments not only in OCD symptoms, but also in depression, anxiety,somatic symptoms, and in becoming extroverted rather than introvertedand withdrawn.
Discussion. In follow-ups of the two cases at 15 and 13 months after
completion of treatment, both patients were maintaining improvementsin OCD symptoms as measured by the Padua Inventory and as externallyvalidated through contacts with family members. Since research has
D. Corydon Hammond is Professor, Physical Medicine & Rehabilitation, Univer-
sity of Utah School of Medicine.
Address correspondence to: D. Corydon Hammond, PhD, Professor, Physical Med-
icine & Rehabilitation, University of Utah School of Medicine, 30 North 1900 East,Salt Lake City, UT 84132-2119 (E-mail:
[email protected]).
The author is appreciative of the generous assistance of Stephen Shepperd, PhD, in
administering the Yale Brown Obsessive Compulsive Scale to these patients' post-treatment.
Journal of Neurotherapy, Vol. 7(2) 2003
2003 by The Haworth Press, Inc. All rights reserved.
JOURNAL OF NEUROTHERAPY
found that pharmacologic treatment of OCD produces only very modestimprovements and behavior therapy utilizing exposure with responseprevention is experienced as quite unpleasant and results in treatmentdropouts, neurofeedback appears to have potential as a new treatmentmodality.
[Article copies available for a fee from The Haworth Document De-livery Service: 1-800-HAWORTH. E-mail address: <docdelivery@haworthpress.
com> Website:
2003 by The HaworthPress, Inc. All rights reserved.]
KEYWORDS. Neurofeedback, EEG, biofeedback, quantitative EEG,
OCD, obsessive-compulsive disorder
Obsessive compulsive disorder (OCD) has a lifetime incidence in the
range of 1% to 3% (Karno, Golding, Sorenson, & Burnam, 1988; Jenike& Brotman, 1984) and is widely regarded as having a strong biologicalbasis. In a qEEG study, Kuskowski et al. (1993) discovered lower abso-lute power in delta, beta 1 and beta 2 bandwidths frontally and in theright hemisphere in OCD patients. They further discovered increasedalpha relative power across temporo-parietal, central and occipital re-gions, along with decreased relative power in beta bands in the left fron-tal region. Their research additionally revealed severe right hemispherehypoactivity, particularly in beta relative power. This is an interestingfinding since treatment with clomipramine has been found to result inincreased right hemisphere activity (MacCrimmon & Arato, 1991),which may help normalize the electroencephalogram (EEG) in OCDpatients with this pattern.
On the surface these findings seem somewhat at odds with most PET
and SPECT studies of OCD which have reported increased frontalblood flow and metabolism in mediofrontal, anterior cingulate, rightfrontal, and/or orbitofrontal areas (Baxter et al., 1987; Baxter, Phelps, &Mazziotta, 1988; Benkelfat et al., 1990; Harris, Pearlson, & Hoehn-Saric, 1993; Machlin, Harris, & Pearlson, 1991; Nordahl et al., 1989;Perani et al., 1995; Piacentini & Bergman, 2000; Rauch, Whalen,Dougherty, & Jenike, 1998; Rubin, Villaneuva-Meyer, & Anath, 1992;Sawle, Hymas, & Lees, 1991; Saxena, Brody, Schwartz, & Baxter,1998; Swedo, Schapiro, & Grady, 1989; Szeszko et al., 1999). Theneuroimaging findings converge in implicating a cortico-striato-thalamo-
cortical network. Resting studies of OCD seem to indicate hyperactivityin the orbitofrontal and anterior cingulate cortex and caudate nucleus,with this being attenuated under conditions of symptom provocation,and which attenuate following successful treatment (Rauch, 2000).
There are some evoked potential studies that are more congruent
with neuroimaging findings. The anterior cingulate cortex may be in-volved with monitoring of behavior (Posner & Rothbart, 1998). Anevent related potential called error-related negativity (ERN) is a waveform that is associated with making mistakes or errors (Gehring, Coles,Meyer, & Donchin, 1990). It reflects the activity of a general error-pro-cessing system and one of the symptoms of OCD consists of excessivechecking, rumination, and doubt–which amount to excessive responsemonitoring. The size of an ERN is sensitive to the size of an error. ERNhas been localized as being generated from a single source in the medialfrontal cortex (Dehaene, Posner, & Tucker, 1994; Gehring, Goss, Coles,Meyer, & Donchin, 1993; Holroyd, Dien, & Coles, 1998; Luu, Collins, &Tucker, 2000). Gehring, Himle, and Nisenson (2000) found that theERN was increased in OCD patients compared with matched controls,and the magnitude of the ERN was related with symptom severity. AnfMRI study (Ursu et al., 2001) documented increased error-related ac-tivity in the anterior cingulate cortex in OCD patients, and this degree ofactivity was likewise correlated with severity of symptoms. SupportingGehring et al. (2000), Hajcak and Simons (2002) also found a signifi-cantly larger Fz maximal negativity was associated with error responsesin college undergraduates with OCD characteristics compared with stu-dents without such characteristics.
Some of the discrepancy between neuroimaging and qEEG studies
may also stem from a bias in neuroimaging studies (Rauch, 2000); theyoften concentrate on only a limited number of brain areas, producing apotential confirmatory bias. Simpson, Tenke, Towey, Liebowitz, andBruder (2000) conducted the first qEEG study under conditions ofsymptom provocation. Importantly, they found that only live exposure(and not imaginal exposure) to contaminants produced significant EEGchanges. This is of importance since some OCD neuroimaging studiesonly used imaginal exposure. Simpson et al. (2000), using only vertexelectrode sites, found a significant shift in the anterior-to-posterior to-pography of alpha power during live exposure compared with a controlcondition. Live exposure was associated with an increase in OCDsymptoms and an increase in posterior relative to anterior alpha. No sig-nificant shifts occurred in the theta or beta bands. The observed changeswere interpreted as reflecting a relative shift in brain activation from

JOURNAL OF NEUROTHERAPY
posterior to anterior, which would be consistent with neuroimagingstudies finding enhanced frontal activation during symptom provoca-tion in OCD.
Other qEEG research has identified two subtypes of OCD patients
(Mas, Prichep, John, & Levine, 1993; Perros, Young, Ritson, Price, &Mann, 1992; Prichep, Mas, & John, 1989; Prichep et al., 1993; Silver-man & Loychik, 1990). Prichep et al. (1993) found one subgroup withexcess alpha throughout most of the brain, but most excessive at T5, P3,O1 (which would coincide with findings by Kuskowski et al., 1993),and the frontal poles, along with a mild excess of beta in frontal, centraland mid-temporal areas. Their other subgroup had a theta excess, mostextreme throughout frontal areas and at posterior temporal electrodes.
Figure 1 displays these subgroups. Theta abnormalities have also beenreported by others (Insel, Donnelly, Lalakea, Alterman, & Murphy,1983; Jenike & Brotman, 1984; Pacella, Polatin, & Nagler, 1944; Rock-well & Simons, 1947).
FIGURE 1. Group Average Topographic Maps (nose up) for Z Relative Powerin the Delta, Theta, Alpha, and Beta Frequency Bands for the Two NeurometricClusters of Patients with Obsessive-Compulsive Disorders
Reprinted from Prichep, L. S., Mas, F., Hollander, E., Liebowitz, M., John, E. R., Almas, M., DeCaria, C. M., & Le-vine, R. H. (1993). Quantitative electroencephalography (QEEG) subtyping of obsessive compulsive disorder. Psy-chiatry Research, 50(1), 25-32, with permission from Elsevier Science.
Delayed onset of mu event-related desynchronization with prepara-
tion for movement and less post-movement beta (20 Hz) synchroniza-tion was reported by Leocani et al. (2001), a finding also found inParkinson's disease (Pfurtscheller, Pichler-Zalaudek, Ortmayr, Kiez, &Reisecker, 1998). Leocani et al. (2001) suggested that a lower level ofbeta synchronization in OCD after a simple, self-paced movementraises a question about whether this may reflect the inability of these pa-tients to inhibit themselves from compulsive actions. In this regard,lower P300 amplitudes in orbitofrontal areas in OCD patients (Malloy,Rasmussen, Braden, & Haier, 1989) likewise suggest impaired inhibi-tory mechanisms. Reduced motor cortical inhibition has also been foundin this population with transcrancial magnetic stimulation (Greenberget al., 2000). Perhaps related to these findings, Flor-Henry, Yeudall,Koles, and Howarth (1979) noted reduced left temporal (T3) variabilityin beta frequencies, but they did not examine frequencies lower than al-pha.
Similar to Prichep, Mas, and John (1989), Prichep et al. (1993) and
Mas, Prichep, John, and Levine (1993), Perros, Young, Ritson, Price,and Mann (1992) noted theta excess in 10 of 13 OCD patients in the6.0-7.5 Hz range, predominantly in the left fronto-temporal area. Theypointed out that such activity is frequently attributed to disturbances indeep midline structures (Gloor, 1976), which would be supported bysome of the neuroimaging studies cited earlier. We might speculate thatthis theta subtype of OCD may be the group which has often producedsome of the neuroimaging findings noted above. Relevant to one of thecase reports which will be presented, another qEEG study has alsopointed to abnormalities in the left posterior temporal area. Silvermanand Loychik (1990) examined three siblings ranging from 23 to 29years of age, all of whom had OCD. The left posterior temporal areawas identified as abnormal in all three, both on qEEG evaluation and onauditory and visual evoked potentials, while the asymptomatic parentswere entirely normal on all measures.
Treatments for OCD
Behavior therapy commonly uses exposure and response prevention
techniques to treat OCD (Foa & Franklin, 2001), with claims that 76%to 86% of patients who complete treatment make improvements. In anearlier review by Foa, Steketee, and Ozarow (1985), they reported thatin over 200 patients, 51% reduced their symptoms at least 70%. InGreist's (1990) review, however, he notes what I have experienced in
JOURNAL OF NEUROTHERAPY
years past using a behavior therapy approach with OCD patients, whichis that the greatest problem is that many patients dislike this treatmentand fully one-quarter decline treatment or sabotage it with overt or co-vert avoidance. He also notes that behavior therapy has proven less suc-cessful with pure obsessional disorder (without rituals) and estimatesthe percent improvement in symptoms experienced following behaviortherapy as 50%. Nonetheless, this psychiatrist's review estimates the de-gree of symptomatic improvement with serotonin drugs as only being30%. Goodman, McDougle, and Price (1992) similarly found thatsymptom amelioration in OCD treatment with serotonin uptake inhibi-tors is about 35% on average, and that 50% of patients experience onlypartial symptomatic improvement.
The mean from four separate samples (Goodman, Price, Rasmussen,
Mazure, Fleischmann et al., 1989; Goodman, Price, Rasmussen, Mazure,Delgado et al., 1989) of OCD patients on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS; Goodman, Price, Rasmussen, Mazure,Fleischmann et al., 1989) is 24.7 (standard deviation = 6). A very recent(Ackerman & Greenland, 2002) meta-analysis of 25 drug studies foundthat with the most effective pharmacologic treatment for OCD (clomi-pramine) that the average drug treatment effect on the Y-BOCS was10.64 (uncorrected for placebo effects), which is a 1.33 standard devia-tion improvement. In fluvoxamine (Prozac) studies, the mean Y-BOCSimprovement was only 5.4 points. Interestingly, they discovered thatthe longer the clomipramine drug trial went on, the less improvementthey found. Thus, 12-week trials had 5.78 less points of improvementthan 10-week trials. Older patients also had less improvement onclomipramine. They found that the longer the pre-randomization period(during which some placebo responders are often dropped from inclu-sion in drug studies), the less improvement in drug response and in pla-cebo response. The reviewers concluded that "the numerous side effectsof clomipramine may have contributed to its greater effect size in pla-cebo comparisons" (p. 315). This same conclusion was reached previ-ously by the same group (Ackerman et al., 1996), as well as byAbramowitz's (1997) review. This is particularly relevant because re-cent reviews of antidepressant drugs studies (Antonuccio, Danton, DeNelsky,Greenberg, & Gordon, 1999; Kirsch & Sapperstein, 1998; Moncrieff,2001) have identified that these studies commonly use inactive place-bos which have no side effects, resulting in many patients and raterscorrectly discerning to which group they have been assigned, essen-tially unblinding the study. However, one review (Thomson, 1982)where an active placebo (e.g., atropine, which causes anticholinergic
side effects) was used found that only one in seven studies identified theantidepressant as superior to placebo. Another similar review (Moncrieff,Wessely, & Hardy, 1998) discovered that in only two of nine studieswas antidepressant medication superior to an active placebo.
Despite the very modest effects of medication, there is evidence that
qEEG has potential to assist in predicting medication response in treat-ing OCD (Prichep et al., 1993). Those patients with excess alpha rela-tive power (with some frontal and central beta excess) were found torespond positively 82% of the time to serotonin mediated antidepres-sants, whereas, the second subtype with increased theta relative power(with some alpha minima) failed to improve 80% of the time withSSRIs. Nonetheless, it must be emphasized that medication treated pa-tients remain dependent on the medication, and in one study (Pato,Zohar-Kadouch, & Zohar, 1988), 89% of patients treated with clomi-pramine (Anafranil) relapsed after discontinuation of medication. Natu-rally, medications such as clomipramine also have numerous sideeffects which must be tolerated such as dry mouth, blurred vision, con-stipation, sweating, sedation, dizziness, and retarded ejaculation.
Psychiatry has also resorted to neurosurgical treatment for OCD, per-
forming cingulotomies in cases that have proven resistant to both medi-cation and a trial of behavior therapy. However, using a somewhatliberal criteria of having produced at least 35% improvement on theYale-Brown Obsessive Compulsive Scale, such psychosurgery has onlybenefited from one-quarter to one-third of patients (Dougherty et al.,2002; Jenike et al., 1991), even with the confound that most of the pa-tients continued receiving pharmacotherapy following cingulotomy.
Rauch (2000) summarized, "For neurosurgical treatment of OCD, theoverall rate of efficacy is quite modest, the costs are high, and the risksare considerable" (p. 169). It is thus apparent that current psychiatrictreatment of OCD has significant limitations.
There also exists interesting neuroimaging research on pre-treatment
brain characteristics that predict successful outcome. What is particu-larly interesting about these studies is that they show biological brainchanges occurring following successful behavior therapy (exposure andresponse prevention), with such changes not occurring in persons whodid not change in treatment or in normal controls. Schwartz, Stoessel,Baxter, Martin, and Phelps (1996) and Baxter et al. (1992) found signif-icant changes in glucose metabolism in the right caudate following suc-cessful behavior therapy. They also found a significant correlationbetween change in left orbital frontal cortex with change in Y-BOCSscores, which has also been found by Swedo et al. (1992) in successful
JOURNAL OF NEUROTHERAPY
medication treatment. Brody et al. (1998) reported normalization of leftorbitofrontal cortex metabolism predicted positive treatment responseto behavior therapy. They found that higher metabolism in the leftorbitofrontal cortex predicted greater improvement with behavior ther-apy, but a worse outcome from fluoxetine treatment.
This paper will report on the treatment of two cases of OCD with
neurofeedback. In both cases following the initial history taking, aquantitative EEG (qEEG) was done to evaluate brain function. Vigi-lance-controlled EEG was digitally recorded from the patients withLexicor NRS-24 equipment with recording electrodes placed accordingto the 19 standard regions defined by the International 10/20 System ofelectrode placement, referenced to linked ears. All electrode impedancelevels were below 3 Kohms, with no interelectrode differences of morethan 500 ohms and ear references which were perfectly balanced. Thevigilance level was controlled by noting signs of drowsiness appearingin the EEG, and then pausing the recording and verbally interactingwith the patients, while they moved their arms and legs in the chair. Abipolar recording channel was used to monitor eye movement artifact.
In each case, approximately 20 minutes of eyes-closed resting EEGwere recorded and edited to reduce artifact. The recordings were ofgood quality. From the digitally stored EEG, 132 seconds and 120 sec-onds of EEG in the two cases were subjected to quantitative spectralanalysis. In the first case, a second sample of 62 seconds was also gath-ered for purposes of establishing test-retest reliability. The R squaredvalue for alpha was 96.5%. The R squared values for single hertz topog-raphies may be seen in Figure 3. The results of spectral analysis from1-32 Hz were displayed as computed color-graduated topographic mapsand compared via a Z-score transformation to age-regressed data basesof normal subjects using the Nx Link database and the Thatcher Life-span database with the NeuroRep Analysis and Report System, the lat-ter of which was used to generate one hertz topographic maps. Thefemale patient's eyes-closed EEG was also analyzed utilizing low reso-lution electromagnetic tomography (LORETA) to provide an estima-tion of the localization of underlying generators of the patient's alphaactivity. Both patients engaged in an informed consent process andsigned informed consent forms. The patients were tested pre-treatmentand post-treatment with the Yale-Brown Obsessive Compulsive Scale
(Goodman, Price, Rasmussen, Mazure, Fleischmann et al., 1989; Good-man, Price, Rasmussen, Mazure, Delgado et al., 1989) and the Padua In-ventory (Burns, Keortge, Formea, & Sternberger, 1996), both of whichdemonstrate good reliability and validity. In the first case, the Minne-sota Multiphasic Personality Inventory (MMPI) was also administeredpre- and post-treatment.
This patient was a single, 25 year-old woman who was employed as
an elementary school teacher. She had been diagnosed as having OCDat age 17 by a psychiatrist, and her previous treatment had consisted ofpharmacologic therapy. Her medications had included Prozac, Klonepin,Zoloft, Anafranil, Haldol, Amitriptaline, Effexor, Serzone, and Xanax.
None of the medications had been very effective. She was on .25 mg ofKlonepin per day at the time of the intake interview, but had been off allmedication for over two weeks prior to her qEEG. No other familymember had been formally diagnosed with OCD, but she indicated thatthere were many individuals in her father's family who exhibitedOCD-like behavior. The MMPI was administered because she also de-scribed significant depression. It confirmed severe depression and anxi-ety, very low ego-strength, introversion and being withdrawn, andintense over-emotionality with an extreme, classic pattern for develop-ing somatic complaints. She previously had two suicide attempts, bothat the age of 17. She also suffered with insomnia, requiring two hours tofall asleep. She engaged in bruxism and experienced considerableanxiety. She had been a straight A student in high school. She starteddating at age 16, but had not done much dating since going to college.
Two years previous to our intake interview she was so overwhelmed byher OCD that she was asked to resign at the end of the year. The yearprevious to seeing me she had not taught because she felt too incapaci-tated by her OCD symptoms. These symptoms particularly focused oncontamination obsessions and washing compulsions, as well as obses-sional rumination about harming herself. She also engaged in a lot ofmental counting and excessive blinking. She had currently been teach-ing again for two months, but she was wondering if she would have toresign before long because she was becoming so overwhelmed by theOCD. Her qEEG results from the Nx Link and Lifespan databases maybe seen in Figures 2 and 3. In Figure 4 you can study her LORETA anal-ysis using a Poisson Maximal Frequency Test for alpha frequency. It lo-calized the left temporal alpha to the superior temporal gyrus in the

JOURNAL OF NEUROTHERAPY
FIGURE 2. Case 1: Quantitative EEG Results from the Nx Link Database
vicinity of Brodmann areas 22 and 42, as well as in the middle temporalgyrus, Brodmann area 39, which is in the area of the angular gyrus.
Following informed consent, we decided that due to the severity of
her depression, our initial focus would be on reducing depression.
Therefore, we utilized my depression protocol (Hammond, 2001a).
This protocol was designed in response to the extensive qEEG andneuroimaging literature (summarized in Davidson, 1998a, 1998b) doc-umenting a robust biological marker for depression which consists ofgreater left frontal alpha activity (inactivation) compared with rightfrontal activity. This protocol is also responsive to parallel research(Davidson, 1992; Heller, Etienne, & Miller, 1995; Heller, Nitschke,Etienne, & Miller, 1997; Isotani et al., 2001; Pizzagalli et al., 2002) con-firming that a frontal asymmetry with greater right frontal beta activa-tion (e.g., Fp2, F4) is associated with anxiety, and with panic disorder

FIGURE 3. Case 1: Single Hertz Magnitude Topographic Maps
SINGLE-BAND MAGNITUDE TOPOGRAPHIES
(Wiedemann et al., 1999). A large proportion of depressed patients alsoexperience anxiety. These latter qEEG studies are also congruent withPET and MRI studies (Abercrombie et al., 1996; Canli, Desmond,Zhao, Glover, & Gabrieli, 1998; Chua, Krams, Toni, Passingham, &Dolan, 1999; Dolan et al., 1996; Dolski et al., 1996; George et al., 1995;

JOURNAL OF NEUROTHERAPY
FIGURE 4. Case 1: LORETA Analysis for Alpha Frequency Band
Naveteur, Roy, Ovelac, & Steinling, 1992; Reivich, Alavi, & Gur,1984; Stapleton et al., 1997; Stewart, Devous, Rush, Lane, & Bonte,1988) and transcrancial Doppler ultrasound studies (Troisi et al., 1999)of anxiety which find more right than left fronto-temporal activity, sug-gesting that the frontal cortex is involved in regulating and restrainingsubcortical limbic structures associated with affect. As a bridge facili-tating understanding between neuroimaging and EEG research, a recentneuroimaging study (Nakamura et al., 1999) demonstrated a positivecorrelation between beta activity (13-30 Hz) and cerebral blood flow.
The patient's treatment began with twenty-one sessions of neuro-
feedback using the Roshi system (Hammond, 2001a) with two referen-tial training sites at Fp1 and F3. The Roshi uses photic stimulation,wherein LED lights embedded in glasses vary in their pulsation, frommoment to moment, pulsing on the peak frequency within the frequency
band being reinforced. The Roshi also has very low feedback latency.
The 30 to 35 minute sessions at Fp1 and F3 used a program called BetaMax for the first half of the session, which reinforced 15-18 Hz while si-multaneously inhibiting theta and alpha frequency bands, and finishedthe last half of the session with a program called SMR Max (inhibitingtheta and alpha, reinforcing 12-15 Hz). Because it was observed that herdominant frequency not infrequently dropped into the delta range dur-ing this training, in the last 12 of these sessions we spent the first 10minutes strictly inhibiting delta (1-4 Hz), followed by the Beta Maxprogram and SMR Max program for 10 to 13 minutes each.
After two sessions of this training she reported feeling more energy
and alertness, and being more social, "which is abnormal for me." Sheindicated that her depression level (0-10) had dropped from a 9 or 10 toa level 7. After 9 sessions the patient noted that her depression was "alot better," her anxiety was less, and her sister had noted that her OCDsymptom of excessive blinking had decreased. After 11 sessions she re-ported that a co-worker had also commented that she was blinking lessand seeming calmer. After twelve sessions she said she had been feeling"pretty up, and I've been having more energy than normal and sleepingbetter too. The anxiety is really controllable now." After 13 sessions shecommented, "I actually haven't felt this good in my life."
Based on her qEEG, our training now shifted and we now began us-
ing Neuropathways EEG equipment. This is a unit that samples at250,000 samples per second and digitally filters the EEG signal, with acommon mode rejection ratio >110 db wideband and >120 db at 60 Hz.
I switched to Neuropathways to focus primarily on inhibiting inappro-priate activity. Two sessions were done inhibiting 19.5-25 Hz at Pz-Czwith a sequential (bipolar) montage, while setting the threshold liber-ally to only mildly reinforce 12-14 Hz. This was followed by anothersession inhibiting 18-25 Hz and mildly reinforcing 12-15 Hz at Cz-Fzfor 20 minutes and then inhibiting 19-25 Hz at Cz-T3 for 15 minutes.
After these sessions she said she was mellower, able to concentrate eas-ier, and "my mind didn't go as fast." We then did four sessions atT3-Cz, inhibiting 19-25 Hz while mildly reinforcing 12-15 Hz. Afterthe first two of these four sessions she reported feeling less preoccupa-tion with food contamination, that her OCD was improving, and she feltminimal depression. Due to some difficulty sleeping, we then did oneRoshi session of SMR Max at C3 and C4. The goal of this session was tohave a calming effect and to also encourage beta spindles and enhancedability to fall asleep.
JOURNAL OF NEUROTHERAPY
Next, we began focusing on the excess alpha activity in the left poste-
rior area. Comparing her qEEG relative power alpha (Figure 2) with thealpha subtype from Prichep et al.'s (1993) research (see Figure 1), youwill note that they are almost identical. It was my hope, therefore, thattraining at this site would begin to seriously impact her OCD symp-toms. For the next eight consecutive sessions we inhibited 6.5-11 Hzwhile mildly reinforcing 15-18 Hz at T5-P3 with a sequential montage,using the Neuropathways unit. As hoped, with each session she re-ported that her OCD symptoms and brooding seemed to be improving.
After seven sessions with this placement she estimated that her obses-sions had improved by 75%. We then returned for two sessions to theleft frontal area (Fp1-F3), using the Roshi depression protocol to rein-force changes in depression. A couple of days following these sessions,I spoke with the father of the patient on the telephone. He told me that afew days earlier his daughter had told him, "Dad, for the first time in mylife I feel normal." Twelve more sessions focused on T5-P3, and in oneof those sessions the time was split between T5-P3 and T5-O1. Two fur-ther sessions to reinforce changes in depression were also done atFp1-F3. During this time she was reporting no problems with either de-pression or OCD symptoms.
After 50 sessions of neurofeedback, another MMPI was adminis-
tered. The striking changes in her two MMPIs may be seen in Figure 5.
Her depression normalized from a severe level and she became muchless withdrawn, going from being introverted to extroverted. These lat-ter changes have been commonly observed by me in pre-post testing us-ing my depression protocol. Increasing left frontal activation would beanticipated to produce such changes, since the left frontal area is associ-ated with not only happy emotions, but also with approach motivation(Davidson, 1998a). Her extreme levels of both over-emotionality andsomatic symptoms decreased to within normal limits. Her anxiety andOCD symptoms, as measured in scales A and 7, dramatically decreasedand her ego-strength and resilience increased. She was defensive in hertest taking set at both administrations of the MMPI. Nonetheless, in herpre-treatment testing she still showed extreme levels of psychologicaldisturbance, and in her post-treatment testing her defensiveness wasonly mildly greater.
At this same time the Padua Inventory was readministered, and I had
a colleague administer the Y-BOCS to alleviate contamination effects.
The last eight sessions of neurofeedback had placements at T5-Pz, in-hibiting 4-8 Hz while mildly reinforcing 15-18 Hz. The first three ofthese maintenance sessions were held at two- to three-week intervals.
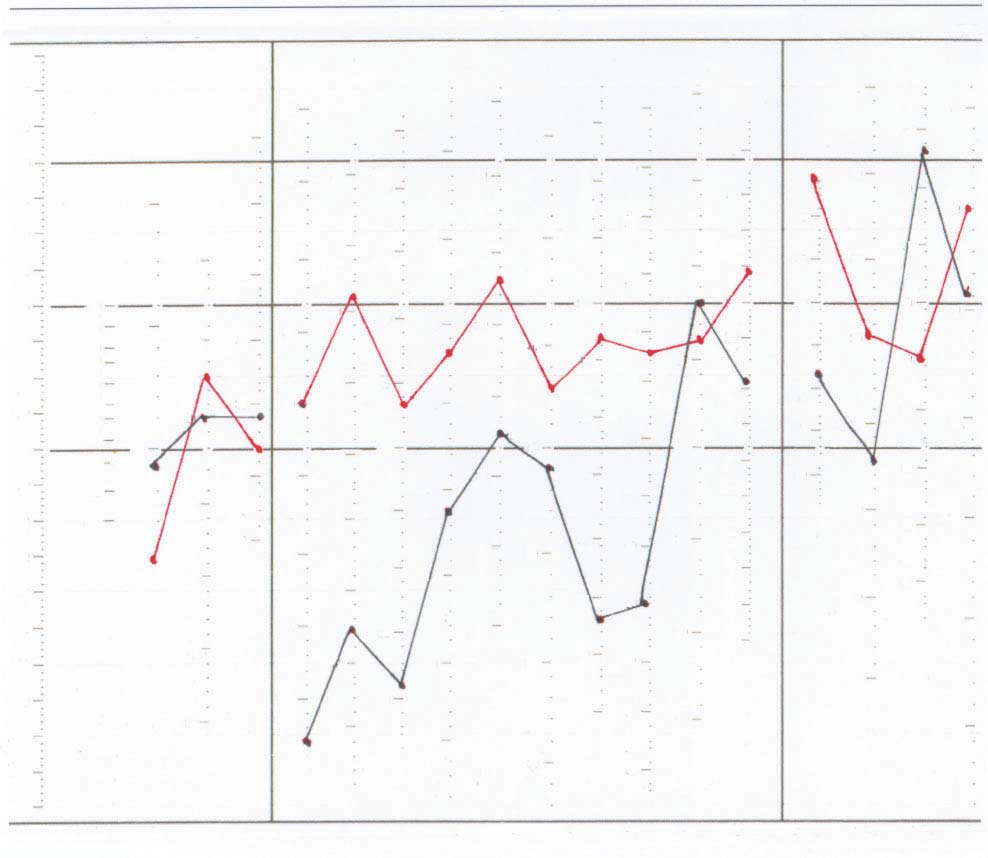
FIGURE 5. Case 1: Pre- and Post-Treatment MMPI
Pt+1K Sc+1K Ma+2K
Pt+1K Sc+1K Ma+2K
The final five sessions were spaced out over a five-month period for fol-low-up reinforcement and to check for maintenance of changes. Sevenand a half months after termination of the five-month maintenancephase of treatment (and 15 months after the main treatment phase), Ispoke with the patient and the Padua Inventory was readministered tothe patient. I had a telephone interview with her mother at about thesame time, obtaining external confirmation of the maintenance ofchanges.
Table 1 shows the changes in this patient on the Y-BOCS and the
Padua Inventory from pre- to post-treatment and on follow-up. TheY-BOCS is the most respected measure of OCD and generally patientsmust score over 16 to be included in medication trials. The patientscored 26 initially, slightly above the averaged mean for four samples ofOCD patients (Goodman, Price, Rasmussen, Mazure, Fleischmann et
JOURNAL OF NEUROTHERAPY
TABLE 1. Case 1: Pre-Post-Follow-Up OCD Outcome Measures
OCD MEAN & S.D.
54.93 (S.D. = 16.72)
PRE-TREATMENT SCORE
al., 1989; Goodman, Price, Rasmussen, Mazure, Delgado et al., 1989)on the Y-BOCS total score. The Y-BOCS (Goodman, Price, Rasmussen,Mazure, Fleischmann et al. 1989) mean for the Obsessions subscale is10.7 and her pre-treatment score was 12.5. The Y-BOCS mean for thecompulsions subscale is 11.2, and she scored 13.5. An independent ex-aminer interviewed her with the Y-BOCS after 50 neurofeedback ses-sions. Instead of 26, she now scored 4–twice the average reduction inscore that is usually found in drug studies with the most effective phar-macologic treatment (Ackerman & Greenland, 2002). This is a 3.67standard deviation improvement, compared with the 1.33 standard de-viation average improvement that results from the most effective phar-macologic treatment. Her score was now 3 on the Obsessions subscale,and 1 on the Compulsions subscale. The reliability on both administra-tions was rated as excellent. The Y-BOCS was not administered in afollow-up interview because the patient had moved away to anothergeographic area. However, in addition to a telephone interview with thepatient and her mother, she did take the self-administered Padua Inven-tory 15 months following the second (post-treatment) administration ofthe YBOCS and the Padua Inventory.
On the Padua Inventory, her pre-treatment score of 72 significantly
exceeded the mean for an OCD population (54.93; SD = 16.72; Burns etal., 1996), placing her one standard deviation above the mean for OCDpatients. After 50 neurofeedback sessions her total score was 8, and onfollow-up 15 months later, her score was 12. These improvements werecomparable to those seen on the Y-BOCS (see Table 1). The mean totalscore on the Padua Inventory for a normal sample is 21.78 (SD = 16.33).
Thus, at the completion of 50 sessions she was almost one standard de-
viation below the mean for a normative sample. On subscale 1 (Obses-sive Thoughts about Harm to Self or Others; OCD Mean = 10.0), herpre-treatment score was 18, her post-treatment score was 4, and her fol-low-up score was 3. On subscale 2 (Obsessive Impulses to Harm Self orOthers; OCD Mean = 6.0), her pre-treatment score was 6, her post-treat-ment score was 0, and her score on follow-up was 1. On subscale 3(Contamination Obsessions and Washing Compulsions; OCD Mean =13.87), her pre-treatment score was 25, her post-treatment score was 3,and her follow-up score was 2. On subscale 4 (Checking Compulsions;OCD Mean = 19.87), her pre-treatment score was 15, her post-treatmentscore was 1, and her score on follow-up was 4. Finally, on subscale 5(Dressing/Grooming Compulsions; OCD Mean = 5.2), her pre-treat-ment score was 8, her post-treatment score was 0, and her score on fol-low-up was 2. Thus, in her 15 month follow-up she scored at or belowthe mean for normal, non-OCD individuals on all Padua Inventorysubscales. This is even more significant because she was feeling underextra stress, having just begun teaching after a summer vacation. Shewas on no medication.
The second case was a 25-year-old male who initially presented as
having problems with attention deficit disorder. He had been "on heavydoses of Ritalin" for years. He indicated that when he began taking it, "itchanged everything" and he felt he could think and function normally.
"I looked forward to using Ritalin," he said, but at the same time it madehim feel stigmatized. He had recently read about neurofeedback andwanted to experiment with its potential. His history included a signifi-cant previous problem with marijuana abuse and a problem with alco-hol abuse. He said, "I can't stay with anything for more than fiveminutes. I want instant gratification." He had also had "breakdowns" inwhich he became depressed and would cry to his parents on the tele-phone. He had previously been on Effexor and Paxil, but was not cur-rently depressed and was only taking Ritalin. He met the diagnosticcriteria for ADD and ADHD. After an intake interview, we gatheredqEEG data after he had been off Ritalin for three days.
In giving him feedback on the analysis, I commented on the mild ex-
cess of beta over the general area of the anterior cingulate gyrus (seeFigure 6, electrode sites Fz and Cz), and inquired about OCD symptom-atology. It quickly became apparent that he had many OCD symptoms(e.g., obsessions with contamination and washing rituals, and checking

JOURNAL OF NEUROTHERAPY
FIGURE 6. Case 2: Quantitative EEG Results from the Nx Link Database
compulsions) and he admitted to feeling he "must be absolutely per-fect," whether it was in playing music or making hamburgers. There-fore, I administered the Y-BOCS and gave him the Padua Inventory totake home. His Y-BOCS score of 25 and Padua Inventory score of 62confirmed the dual diagnosis of OCD.
Forty-four 30-minute long sessions were spent inhibiting 19-25 Hz
beta, while mildly reinforcing 12-15 Hz, typically for 15 to 20 minutesat Fz-Cz (using a sequential montage with a Neuropathways neuro-feedback unit), followed by 15 minutes with the same protocol atCz-C4. Interestingly, after two sessions he indicated: "A lot has changed.
For two days I have felt I was no longer a prisoner to any situation. I feelturned-on, in a non-sexual way." He further volunteered that he hadtended to compulsively masturbate and had also used masturbation as asoporific. However, he had been feeling no compulsion to masturbate.
He also felt that he tracked words more smoothly on a page in reading.
Since there had been only two sessions, this could be the result of posi-tive expectancy, but on the other hand, medication responses had notbeen overly positive with antidepressants. After four sessions he re-ported sleeping much better. After six sessions, he indicated that twofriends, his father, and a sister had all spontaneously indicated that heseemed calmer. He said, "There's an absence of rudimentary fear andparanoia." After another session, he described himself as "mellow," andsaid, "It's much easier for people to be around me. Women actuallywant to get to know me!" After still another session, he indicated he was"in a good mood all the time." Following nine sessions he said that theRitalin he was taking had been feeling more and more potent, and,therefore, he had been cutting it down and was now taking only betweenone-quarter and one-eighth of the prescribed dose daily.
At this time we began occasionally spending the first half of the ses-
sion inhibiting 20-25 Hz beta at Cz-C4 or Fz-Cz, and then moving theelectrodes to F7 and F8 and inhibiting 7-11 Hz while mildly reinforcing13-16 Hz. This was done to begin addressing his ADD problems fur-ther, since F7 and F8 were both areas of alpha excess on his qEEG, asmay be seen in Figure 6. After twelve sessions he said that his visualtracking of words felt "improved immensely." He also indicated, "Ican't even drink coffee anymore" because rather than helping his con-centration as it did before, it caused him to feel over stimulated. Afteranother session he commented that his concentration was better whenhe was reading, and he was having fewer intrusive thoughts. Followinghis fifteenth session he reported being able to read for long periods oftime. He continued to steadily report feeling better. After 26 sessions heindicated that his memory with reading was improved, he had less so-cial anxiety, felt more energetic, and found he could do things musicallythat he could not do before due to anxiety.
As treatment progressed, we also did some prefrontal training at Fp1
and Fp2, inhibiting 7-11 Hz and mildly reinforcing 12.5-15 Hz. The pa-tient felt that this frontal training improved his concentration "a lot,"helped him to feel more confident and less afraid socially, and he be-lieved that it felt as if it also assisted with the OCD symptoms. By thetime we had reached 50 total sessions, he was feeling excellent. Womenhe dated found him much more mature and easier to relate to, and hisfamily found him to be much easier to get along with. OCD symptomswere minimal. After 56 thirty-minute sessions, 35 of which had focusedon inhibiting beta in the Fz-Cz-C4 area, an independent colleague re-administered the Y-BOCS. His score had now dropped from 25 to 10–adecrease of 2.5 standard deviations.
JOURNAL OF NEUROTHERAPY
The patient was feeling a desire to enhance his academic abilities fur-
ther in anticipation of returning to college, and he had the financialresources to do so. Therefore, we continued neurofeedback. At the con-clusion of treatment (93 total sessions), he had gone through 44 sessionsinhibiting beta over the Fz-Cz-C4 area, 22 sessions inhibiting alpha andreinforcing low beta frequencies at F7-F8, 21 sessions at Fp1-Fp2, threeand a half sessions inhibiting 2-9 Hz at O1-O2, and two and a half ses-sions inhibiting alpha in the parietal area.
A summary of the pre-, post-, and follow-up testing on this case is
found in Table 2. At the end of treatment, after the 44 thirty-minute ses-sions of inhibiting beta along the vertex (and 93 total sessions), theY-BOCS was again administered and his score had decreased furtherfrom 10 to 7. This translates to an improvement of 3 standard deviationsin his Y-BOCS score from the beginning of treatment. His score on thePadua Inventory at that time had dropped from 62 to 7, representing a3.4 standard deviation improvement from his pre-treatment level. Atthe conclusion of treatment, the patient moved out of state. However, 13months later I was able to speak with his sister who had just returnedfrom an extended visit with him. She reported that he remained dramati-cally changed from his pre-treatment adjustment. I also interviewedhim on the telephone and had him complete the Padua Inventory. Hehad always had anxiety about flying, and since the September 11, 2001terrorist attack in New York City, his fear had been exacerbated, but heno longer experienced any OCD symptoms. He was not on any medica-tion for OCD and was not taking Ritalin. Nonetheless, he said, "Myconcentration is still a million times better." The improvement in con-centration and OCD had given him the confidence to return to college.
His 13-month follow-up score on the Padua Inventory was now 5, a 3.4
TABLE 2. Case 2: Pre-Post-Follow-Up OCD Outcome Measures
OCD MEAN & S.D.
54.93 (S.D. = 16.72)
PRE-TREATMENT SCORE
standard deviation improvement from his pre-treatment score. HisPadua Inventory subscale 1 (Obsessive Thoughts about Harm to Self orOthers; OCD Mean = 10.0) pre-treatment score was 15, his post-treatmentscore was 2, and his follow-up score was 2. On subscale 2 (ObsessiveImpulses to Harm Self or Others; OCD Mean = 6.0), his pre-treatmentscore was 2, his post-treatment score was 1, and his score on follow-upwas 0. On subscale 3 (Contamination Obsessions and Washing Com-pulsions; OCD Mean = 13.87), his pre-treatment score was 20, hispost-treatment score was 2, and his follow-up score was 2. On subscale4 (Checking Compulsions; OCD Mean = 19.87), his pre-treatmentscore was 19, his post-treatment score was 2, and his score on follow-upwas 1. Finally, on subscale 5 (Dressing/Grooming Compulsions; OCDMean = 5.2), his pre-treatment score was 6, his post-treatment scorewas 0, and his score on follow-up was 0. Thus, in his 13 month fol-low-up he scored at or below the mean for normal, non-OCD individu-als on all Padua Inventory subscales.
SUMMARY AND CONCLUSIONS
In research with uncontrolled epilepsy (summarized in Sterman,
2000, which has included placebo-controlled, blinded studies) neuro-feedback has proven capable of reconditioning brain wave patterns.
Outcome research has also been done on neurofeedback with ADD/ADHD, learning disabilities, depression, anxiety, brain injury, fibro-myalgia, and posttraumatic stress disorder (Hammond, 2001b). This isthe first publication, however, on the treatment of obsessive-compul-sive disorder with neurofeedback. Quantitative EEGs were gathered ontwo consecutive OCD patients seeking treatment. This assessment thenguided individualized protocol selection for subsequent neurofeedbacktraining. The qEEG findings in the first case almost identically matchedthe average profile of an alpha subtype of OCD, leading to treatmentfocused in the left posterior area after we alleviated her depression. Theauthor had never heard of anyone using this neurofeedback protocoland would not have considered using it without being guided by a qEEGassessment. The patient's more significant OCD symptoms primarilychanged following treatment focused in the left posterior area. Scoreson the Yale-Brown Obsessive-Compulsive Scale and the Padua Inven-tory normalized following neurofeedback. An MMPI was administeredpre-post to the first patient, who showed dramatic improvements in notonly OCD symptoms, but also in depression, anxiety, somatic symp-
JOURNAL OF NEUROTHERAPY
toms, and in becoming extroverted rather than introverted and with-drawn. In follow-ups of the two cases at 15 and 13 months aftercompletion of treatment, both patients were maintaining improvementsin OCD symptoms as measured by the Padua Inventory and as exter-nally validated through contacts with family members.
Since research has found that pharmacologic treatment of OCD pro-
duces only very modest improvements, and behavior therapy utilizingexposure with response prevention is experienced as quite unpleasantand results in treatment dropouts, neurofeedback appears to have poten-tial as a new treatment modality for OCD. Further controlled researchshould be pursued in this area.
Abercrombie, H. C., Larson, C. L., Ward, R. T., Schaefer, S. M., Holden, J. E.,
Perlman, S. B., et al. (1996). Metabolic rate in the amygdala predicts negative affectand depression severity in depressed patients: A FDG-PET study. Neuroimage, 3(2), S217.
Abramowitz, J. (1997). Effectiveness of psychological and pharmacological treat-
ments for obsessive-compulsive disorder: A quantitative review. Journal of Con-sulting & Clinical Psychology, 65, 44-52.
Ackerman, D. L., & Greenland, S. (2002). Multivariate meta-analysis of controlled
drug studies for obsessive-compulsive disorder. Journal of Clinical Psychopharm-acology, 22 (3), 309-317.
Ackerman, D. L., Greenland, S., Bystritsky, A., & Katz, R. J. (1996). Relationship be-
tween early side effects and therapeutic effects of clomipramine therapy in OCD.
Journal of Clinical Psychopharmacology, 16, 324-328.
Antonuccio, D. O., Danton, W. G., DeNelsky, G. Y., Greenberg, R. P., & Gordon, J. S.
(1999). Raising questions about antidepressants. Psychotherapy & Psychosomatics,68, 3-14.
Baxter, L. R., Schwartz, J. M., Mazziotta, J. C., Phelps, M. E., Pahl, J. J., Guze, B. H., et
al. (1988). Cerebral glucose metabolic rates in non-depressed patients with obses-sive-compulsive disorder. American Journal of Psychiatry, 145, 1560-1563.
Baxter, L., Phelps, M., Mazziotta, J., Guze, B. H., Schwartz, J. M., & Selin, C. (1987).
Local cerebral glucose metabolic rates in obsessive-compulsive disorder. Archivesof General Psychiatry, 44, 211-218.
Baxter, L., Schwartz, J. M., Bergman, K. S., Szuba, M. P., Guze, B. H., Mazziotta, J.
C., et al. (1992). Caudate glucose metabolic rate changes with both drug and behav-ior therapy for obsessive-compulsive disorder. Archives of General Psychiatry, 49,681-688.
Benkelfat, C., Phelps, M., Mazziotta, J., Guze, B. H., Schwartz, J. M., & Selin, R. M.
(1990). Local cerebral glucose metabolic rates in obsessive-compulsive disorderpatients treated with clomipramine. Archives of General Psychiatry, 147, 846-848.
Brody, A. L., Saxena, S., Schwartz, J. M., Stoessel, P. W., Maidment, K., Phelps, M.
E., et al. (1998). FDG-PET predictors of response to behavioral therapy andpharmacotherapy in obsessive compulsive disorder. Psychiatry Research, 84, 1-6.
Burns, G. L., Keortge, S., Formea, G., & Sternberger, L. (1996). Revision of the Padua
Inventory of obsessive-compulsive symptoms: Distinctions between worry, obses-sions, and compulsions. Behaviour Research & Therapy, 34, 163-173.
Canli, T., Desmond, J. E., Zhao, Z., Glover, G., & Gabrieli, J. D. (1998). Hemispheric
asymmetry for emotional stimuli detected with fMRI. Neuroreport, 9, 3233-3239.
Chua, P., Krams, M., Toni, I., Passingham, R., & Dolan, R. (1999). A functional anat-
omy of anticipatory anxiety. Neuroimage, 9, 563-571.
Davidson, R. J. (1992). Emotion and affective style: Hemispheric substrates. Psycho-
logical Science, 3, 39-43.
Davidson, R. J. (1998a). Affective style and affective disorders: Perspectives from af-
fective neuroscience. Cognition & Emotion, 12, 307-320.
Davidson, R. J. (1998b). Anterior electrophysiological asymmetries, emotion, and depres-
sion: Conceptual and methodological conundrums. Psychophysiology, 35, 607-614.
Dehaene, S., Posner, M. I., & Tucker, D. M. (1994). Localization of a neural system for
error detection and compensation. Psychological Science, 5, 303-305.
Dolan, R. J., Fletcher, P., Morris, J., Kapur, N., Deakin, J. F., & Frith, C. D. (1996).
Neural activation during covert processing of positive emotional facial expressions.
Neuroimage, 4, 194-200.
Dolski, I. V., Malmstadt, J. R., Schaefer, S. M., Larson, C. L., Abercrombie, H. C.,
Ward, R. T., et al. (1996). EEG-defined left versus right frontally activated groupsdiffer in metabolic asymmetry in the amygdala. Psychophysiology, 33, S35.
Dougherty, D. D., Baer, L., Cosgrove, G. R., Cassem, E. H., Price, B. H., Nierenberg,
A. A., et al. (2002). Prospective long-term follow-up of 44 patients who receivedcingulotomy for treatment-refractory obsessive-compulsive disorder. AmericanJournal of Psychiatry, 159 (2), 269-275.
Flor-Henry, P., Yeudall, L., Koles, Z., & Howarth, B. (1979). Neuropsychological and
power spectral EEG investigations of the obsessive-compulsive subjects. Biologi-cal Psychiatry, 14, 119-130.
Foa, E. B., Steketee, G. S., & Ozarow, B. J. (1985). Behavior therapy with obses-
sive-compulsives: From theory to treatment. In M. Mavissakalian, S. M. Turner, &L. Michelson (Eds.), Obsessive-Compulsive Disorder: Psychological and pharma-cological treatment (pp. 49-129). New York: Plenum Press.
Foa, E. B., & Franklin, M. E. (2001). Obsessive-compulsive disorder. In D. H. Barlow
(Ed.), Clinical handbook of psychological disorders (3rd ed., pp. 209-263). NewYork: Guilford.
Gehring, W. J., Coles, M. G. H., Meyer, D. E., & Donchin, E. (1990). The error-related
negativity: An event-related brain potential accompanying errors. Psychophysi-ology, 27, S34.
Gehring, W. J., Goss, B., Coles, M. G. H., Meyer, D. E., & Donchin, E. (1993). A neu-
ral system for error detection and compensation. Psychological Science, 4, 385-390.
Gehring, W. J., Himle, J., & Nisenson, L. G. (2000). Action-monitoring dysfunction in
obsessive-compulsive disorder. Psychological Science, 11, 1-6.
JOURNAL OF NEUROTHERAPY
George, M. S., Ketter, T. A., Parekh, P. I., Horwitz, B., Herscovitch, P., & Post, R. M.
(1995). Brain activity during transient sadness and happiness in healthy women.
American Journal of Psychiatry, 152, 341-351.
Gloor, P. (1976). Generalized and widespread paroxysmal abnormalities. In A. Redmond
(Ed.), Handbook of Electroencephalography & Clinical Neurophysiology, Volume132, Part B. Amsterdam: Elsevier.
Goodman, W. K., McDougle, C. J., & Price, L. H. (1992). Pharmacotherapy of obses-
sive compulsive disorder. Journal of Clinical Psychiatry, 53(Suppl.), 29-37.
Goodman, W. K., Price, L. H., Rasmussen, S. A., Mazure, C., Delgado, P., Heninger,
G. R., et al. (1989). The Yale-Brown Obsessive Compulsive Scale. II. Validity. Ar-chives of General Psychiatry, 46, 1012-1016.
Goodman, W. K., Price, L. H., Rasmussen, S. A., Mazure, C., Fleischmann, R. L., Hill,
C. L., et al. (1989). The Yale-Brown Obsessive Compulsive Scale. I. Development,use, and reliability. Archives of General Psychiatry, 46, 1006-1011.
Greist, J. H. (1990). Treatment of obsessive compulsive disorder: Psychotherapies,
drugs, and other somatic treatment. Journal of Clinical Psychiatry, 51 (8), 44-50.
Greenberg, B. D., Ziemann, U., Cora-Locatelli, G., Harmon, A., Murphy, D. L., Keel,
J. C., et al. (2000). Altered cortical excitability in obsessive-compulsive disorder.
Neurology, 54, 142-147.
Hammond, D. C. (2001a). Neurofeedback treatment of depression with the Roshi.
Journal of Neurotherapy, 4 (2), 45-56.
Hammond, D. C. (2001b). Comprehensive neurofeedback bibliography. Journal of
Neurotherapy, 5 (1-2), 113-128.
Hajcak, G., & Simons, R. F. (2002). Error-related brain activity in obsessive-compul-
sive undergraduates. Psychiatry Research, 110, 63-72.
Harris, G. J., Pearlson, G. D., & Hoehn-Saric, R. (1993). Single photon emission com-
puted tomography in obsessive-compulsive disorder. Archives of General Psychia-try, 50 (6), 498-501.
Heller, W., Etienne, M. A., & Miller, G. A. (1995). Patterns of perceptual asymmetry
in depression and anxiety: Implications for neuropsychological models of emotionand psychopathology. Journal of Abnormal Psychology, 104, 327-333.
Heller, W., Nitschke, J. B., Etienne, M. A., & Miller, G. A. (1997). Patterns of regional
brain activity differentiate types of anxiety. Journal of Abnormal Psychology, 106(3), 376-385.
Holroyd, C. B., Dien, J., & Coles, M. G. H. (1998). Error-related scalp potentials elic-
ited by hand and foot movements: Evidence for an output-independent error-pro-cessing system in humans. Neuroscience Letters, 242, 65-68.
Insel, T. R., Donnelly, E. R., Lalakea, M. L., Alterman, I. S., & Murphy, D. L. (1983).
Neurological and neuropsychological studies of patients with obsessive-compul-sive disorder. Biological Psychiatry, 18, 741-751.
Isotani, T., Tanaka, H., Lehmann, D., Pascual-Marqui, R. D., Kochi, K., Saito, N., et al.
(2001). Source localization of EEG activity during hypnotically induced anxietyand relaxation. International Journal of Psychophysiology, 41, 143-153.
Jenike, M. A., Baer, L., Ballantine, T., Martuza, R. L., Tynes, S., Giriunas, I., et al.
(1991). Cingulotomy for refractory obsessive-compulsive disorder: A long-termfollow-up of 33 patients. Archives of General Psychiatry, 48, 548-555.
Jenike, M. A., & Brotman, A. W. (1984). The EEG in obsessive-compulsive disorder.
Journal of Clinical Psychiatry, 45, 122-124.
Karno, M., Golding, J. M., Sorenson, S. B., & Burnam, M. A. (1988). The epidemiol-
ogy of obsessive-compulsive disorder in five U.S. communities. Archives of Gen-eral Psychiatry, 45, 1094-1099.
Kirsch, I., & Sapperstein, G. (1998). Listening to Prozac, but hearing placebo? A
meta-analysis of antidepressant medication. Prevention & Treatment, 1, 0002a. (Apeer-reviewed APA journal available at volume1/pre0010002a.html)
Kuskowski, M., Malone, S., Kim, S., Dysken, M., Okaya, A., & Christensen, K.
(1993). Quantitative EEG in obsessive compulsive disorder. Biological Psychiatry,33, 423-430.
Leocani, L., Locatelli, M., Bellodi, L., Fornara, C., Henin, M., Magnani, et al. (2001).
Abnormal pattern of cortical activation associated with voluntary movement in ob-sessive-compulsive disorder: An EEG study. American Journal of Psychiatry, 158(1), 140-142.
Luu, P., Collins, P., & Tucker, D. M. (2000). Mood, personality, and self-monitoring:
Negative affect and emotionality in relation to frontal lobe mechanisms of errormonitoring. Journal of Experimental Psychology: General, 129, 43-60.
Machlin, S. R., Harris, G. J., & Pearlson, G. D. (1991). Elevated medial-frontal cere-
bral blood flow in obsessive-compulsive patients: A SPECT study. American Jour-nal of Psychiatry, 148, 1240-1242.
MacCrimmon, D. J., & Arato, H. (1991). Interhemispheric serotonergic asymmetry re-
flected in topographic pharmaco-EEG. Psychiatry Research: Neuroimaging, 40(1), 91-93.
Malloy, P., Rasmussen, S., Braden, W., & Haier, R. J. (1989). Topographic evoked po-
tential mapping in obsessive-compulsive disorders: Evidence of frontal lobe dys-function. Psychiatry Research, 28(1), 63-71.
Mas, F., Prichep, L. S., John, E. R., & Levine, R. (1993). Neurometric Q-EEG sub-
typing of obsessive compulsive disorders. In K. Maurer (Ed.), Imagining of thebrain in psychiatry and related fields (pp. 277-280). Heidelberg, Berlin, Germany:Springer-Verlag.
Moncrieff, J. (2001). Are antidepressants overrated? A review of methodological prob-
lems in antidepressant trials. Journal of Nervous & Mental Disease, 189 (5),288-295.
Moncrieff, J., Wessely, S., & Hardy, R. (1998). Meta-analysis of trials comparing anti-
depressants with active placebos. British Journal of Psychiatry, 172, 227-231.
Nakamura, S., Sadato, N., Oohashi, T., Nishina, E., Fuwamoto, Y., & Yonekura, Y.
(1999). Analysis of music-brain interaction with simultaneous measurement of re-gional cerebral blood flow and electroencephalogram beta rhythm in human sub-jects. Neuroscience Letters, 275 (3), 222-226.
Naveteur, J., Roy, J. C., Ovelac, E., & Steinling, M. (1992). Anxiety, emotion and cere-
bral blood flow. International Journal of Psychophysiology, 13, 137-146.
Nordahl, T. E., Benkelfat, C., Semple, W. E., Gross, M., King, A. C., & Cohen, R. M.
(1989). Cerebral glucose metabolic rates in obsessive-compulsive disorder. Neuro-psychopharmacology, 2, 23-28.
JOURNAL OF NEUROTHERAPY
Pacella, B. L., Polatin, P., & Nagler, S. H. (1944). Clinical and EEG studies in obses-
sive-compulsive disorder. American Journal of Psychiatry, 100, 830-838.
Pato, M., Zohar-Kadouch, R., & Zohar, J. (1988). Return of symptoms after discon-
tinuation of clomipramine in patients with obsessive compulsive disorder. Ameri-can Journal of Psychiatry, 145, 1521-1525.
Perani, D., Colombo, C., Bressi, S., Bonfanti, A., Grassi, F., Scarone, S., et al. (1995).
18F]FDG PET study in obsessive-compulsive disorder: A clinical/metabolic corre-lation study after treatment. British Journal of Psychiatry, 156, 244-250.
Perros, R., Young, E., Ritson, J., Price, G., & Mann, P. (1992). Power spectral EEG
analysis and EEG variability in obsessive-compulsive disorder. Brain Topography,4 (3), 187-192.
Pfurtscheller, G., Pichler-Zalaudek, K., Ortmayr, B., Kiez, J., & Reisecker, F. (1998).
Journal of Clinical Neurophysiology, 15, 243-250.
Piacentini, J., & Bergman, R. L. (2000). Obsessive-compulsive disorder in children.
Psychiatric Clinics in North America, 23 (3), 519-533.
Pizzagalli, D. A., Nitschke, J. B., Oakes, T. R., Hendrick, A. M., Horras, K. A., Larson,
C. L., et al. (2002). Brain electrical tomography in depression: The importance ofsymptom severity, anxiety, and melancholic features. Biological Psychiatry, 52,73-85.
Posner, M. I., & Rothbart, M. K. (1998). Attention, self-regulation and consciousness.
Philosophical Transitions of the Royal Society of London Series B-Biological Sci-ences, 353, 1-13.
Prichep, L. S., Mas, F., & John, E. R. (1989). Neurometric subtyping of obsessive com-
pulsive disorders in psychiatry: A world perspective. Chapter in C. N. Stefanis, A.
D. Rabavilas, & C. R. Soldatos (Eds.), Proceedings of the VIII World Congress ofPsychiatry, Athens, October 12-19, 1989 (pp. 557-562). New York: Elsevier Sci-ence.
Prichep, L. S., Mas, F., Hollander, E., Liebowitz, M., John, E. R., Alman, M., et al.
(1993). Quantitative electroencephalography (QEEG) subtyping of obsessive com-pulsive disorder. Psychiatry Research, 50 (1), 25-32.
Rauch, S. L. (2000). Neuroimaging research and the neurobiology of obsessive-com-
pulsive disorder: Where do we go from here? Biological Psychiatry, 47, 168-170.
Rauch, S. L., Whalen, P. J., Dougherty, D., & Jenike, M. A. (1998). Neurobiologic
models of obsessive-compulsive disorder. In M. A. Jenike, L. Baer, & W. E.
Minichiello (Eds.), Obsessive-compulsive disorders: Practical management(pp. 222-253). St. Louis: Mosby.
Reivich, M., Alavi, A., & Gur, R. C. (1984). Positron emission tomographic studies of
perceptual tasks. Annals of Neurology, 15, (Suppl.), S61-S65.
Rockwell, F. V., & Simons, D. J. (1947). The electroencephalogram and personality
organization in the obsessive compulsive reactions. Archives of Neurology & Psy-chiatry, 57, 71-80.
Rubin, R. T., Villaneuva-Meyer, J., & Anath, J. (1992). Regional 133Xe cerebral blood
flow and cerebral 99m-HMPAO uptake in unmedicated obsessive-compulsive dis-order patients and matched normal control subjects: Determination by high-resolu-tion single-photon emission computed tomography. Archives of General Psychiatry,49, 695-702.
Sawle, G. V., Hymas, N. F., & Lees, A. J. (1991). Obsessive slowness: Functional
studies with positron emission tomography. Brain, 114, 2191-2202.
Saxena, S., Brody, A. L., Schwartz, J. M., & Baxter, L. R. (1998). Neuroimaging and
frontal-subcortical circuitry in obsessive-compulsive disorder. British Journal ofPsychiatry (Supplement), 35, 26-38.
Schwartz, J. M., Stoessel, P. W., Baxter, L. R., Martin, K. M., & Phelps, M. E. (1996).
Systematic changes in cerebral glucose metabolic rate after successful behaviormodification treatment of obsessive-compulsive disorder. Archives of General Psy-chiatry, 53, 109-113.
Silverman, J. S., & Loychik, S. G. (1990). Brain-mapping abnormalities in a family
with three obsessive-compulsive children. Journal of Neuropsychiatry & ClinicalNeurosciences, 2, 319-322.
Simpson, H. B., Tenke, C. E., Towey, J. B., Liebowitz, M. R., & Bruder, G. E. (2000).
Symptom provocation alters behavioral ratings and brain electrical activity in ob-sessive-compulsive disorder: A preliminary study. Psychiatry Research, 95(2),149-155.
Stapleton, J. M., Morgan, M. J., Liu, X., Yung, B. C., Phillips, R. L., Wong, D. F., et al.
(1997). Cerebral glucose utilization is reduced in second test session. Journal of Ce-rebral Blood Flow & Metabolism, 17, 704-712.
Sterman, M. B. (2000). Basic concepts and clinical findings in the treatment of seizure
disorders with EEG operant conditioning. Clinical Electroencephalography, 31(1), 45-55.
Stewart, R. S., Devous, M. D., Rush, A. J., Lane, L., & Bonte, F. J. (1988). Cerebral
blood flow changes during sodium-lactate-induced panic attacks. American Jour-nal of Psychiatry, 145, 442-449.
Swedo, S. E., Pletrini, P., Leonard, H. L., Schapiro, M. G., Rettew, D. C., Goldberger,
E. L., et al. (1992). Cerebral glucose metabolism in childhood-onset obsessive-compulsive disorder: Revisualization during pharmacology. Archives of GeneralPsychiatry, 49, 690-694.
Swedo, S. E., Schapiro, M. G., & Grady, C. L. (1989). Cerebral glucose metabolism in
childhood onset obsessive-compulsive disorder. Archives of General Psychiatry,46, 518-523.
Szeszko, P. R., Robinson, D., Alvir, J. M., Bilder, R. M., Lencz, T., Ashtari, M., et al.
(1999). Orbital frontal and amygdala volume reductions in obsessive-compulsivedisorder. Archives of General Psychiatry, 56 (10), 913-919.
Thomson, R. (1982). Side effects and placebo amplification. British Journal of Psychi-
atry, 140, 64-68.
Troisi, E., Silvestrini, M., Matteis, M., Monaldo, B. C., Vernieri, F., & Caltagirone, C.
(1999). Emotion-related cerebral asymmetry: Hemodynamics measured by func-tional ultrasound. Journal of Neurology, 246, 1172-1176.
Ursu, S., van Veen, V., Siegle, G., MacDonald, A., Stenger, A., & Carter, C. (2001,
March). Executive control and self-evaluation in obsessive-compulsive disorder:An event-related fMRI study. Poster presented at the Cognitive Neuroscience Soci-ety Meeting, New York, Cited in Hajcak & Simons, 2002.
JOURNAL OF NEUROTHERAPY
Wiedemann, G., Pauli, P., Dengler, W., Lutzenberger, W., Birbaumer, N., & Buck-
kremer, G. (1999). Frontal brain asymmetry as a biological substrate of emotions inpatients with panic disorders. Archives of General Psychiatry, 56, 78-84.
RECEIVED: 09/11/02
REVISED: 09/20/02
ACCEPTED: 09/25/02
For FACULTY/PROFESSIONALS with journal subscription
recommendation authority for their institutional library . .
If you have read a reprint or photocopy of this article, would you like to
make sure that your library also subscribes to this journal? If you have
the authority to recommend subscriptions to your library, we will send you
a free complete (print edition) sample copy for review with your librarian.
1. Fill out the form below and make sure that you type or write out clearly both the name
of the journal and your own name and address. Or send your request via e-mail [email protected] including in the subject line "Sample Copy Request"and the title of this journal.
2. Make sure to include your name and complete postal mailing address as well as your
institutional/agency library name in the text of your e-mail.
[Please note: we cannot mail specific journal samples, such as the issue in which a specific article appears.
Sample issues are provided with the hope that you might review a possible subscription/e-subscription withyour institution's librarian. There is no charge for an institution/campus-wide electronic subscriptionconcurrent with the archival print edition subscription.]
Please send me a complimentary sample of this journal:
(please write complete journal title here–do not leave blank)
I will show this journal to our institutional or agency library for a possible subscription.
Institution/Agency Library:
Return to: Sample Copy Department, The Haworth Press, Inc.,
10 Alice Street, Binghamton, NY 13904-1580
Source: http://www.galim.org/files/pdf/HammondOCD.pdf
of care for Future of hepatitis C care in Think Tank hepatitis C care in the Netherlands under guidance of The Argumentation Factory This report was created by: The Argumentation Factory ContentDr Maaike de Vries Drs Silvie Zonderland DesignWillem van den Goorbergh Sponsored byAbbVie How did this report
PODER JUDICIAL DE LA NACIÓN CÁMARA NACIONAL ELECTORAL LEY DE CIUDADANÍA ARGENTINA Ley 26.774 Modifícanse Leyes N° 346, 17.671, 19.945, 23.298, 25.432, 26.215 y 26.571. Sancionada: Octubre 31 de 2012. Promulgada: Noviembre 1 de 2012. El Senado y Cámara de Diputados de la Nación Argentina reunidos en Congreso, etc. sancionan con










