
Cialis ist bekannt für seine lange Wirkdauer von bis zu 36 Stunden. Dadurch unterscheidet es sich deutlich von Viagra. Viele Schweizer vergleichen daher Preise und schauen nach Angeboten unter dem Begriff cialis generika schweiz, da Generika erschwinglicher sind.
Sjzsyj.org

NEURAL REGENERATION RESEARCH
June 2016,Volume 11,Issue 6
Rosiglitazone ameliorates diffuse axonal injury by
reducing loss of tau and up-regulating caveolin-1
expression
Yong-lin Zhao, Jin-ning Song*, Xu-dong Ma, Bin-fei Zhang, Dan-dong Li, Hong-gang Pang
Department of Neurosurgery, the First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi Province, China
How to cite this article: Zhao YL, Song JN, Ma XD, Zhang BF, Li DD, Pang HG (2016) Rosiglitazone ameliorates diffuse axonal injury by re-
ducing loss of tau and up-regulating caveolin-1 expression. Neural Regen Res 11(6):944-950.
Funding: This study was funded by the New Century Supporting Programs to Excellent Talents in China, No. NCET-05-0831.
Protective effects and mechanisms of rosiglitazone against diffuse axonal injury
Jin-ning Song, M.D., [email protected].
Intraperitoneal injection
of rosiglitazone for 3
Rosiglitazone could induce neuroprotection
by attenuating amyloid beta-precusor protein
and hyperphosphorylated tau at Ser404 site and
decreasing the loss of total tau level
Diffuse axonal injury device Diffuse axonal injury rats
after diffuse axonal injury.
Abstract
Rosiglitazone up-regulates caveolin-1 levels and has neuroprotective effects in both chronic and acute brain injury. Therefore, we postu-
lated that rosiglitazone may ameliorate diffuse axonal injury via its ability to up-regulate caveolin-1, inhibit expression of amyloid-beta
precursor protein, and reduce the loss and abnormal phosphorylation of tau. In the present study, intraperitoneal injection of rosiglitazone
significantly reduced the levels of amyloid-beta precursor protein and hyperphosphorylated tau (phosphorylated at Ser404 (p-tau (S404)),
and it increased the expression of total tau and caveolin-1 in the rat cortex. Our results show that rosiglitazone inhibits the expression of amyloid-beta precursor protein and lowers p-tau (S404) levels, and it reduces the loss of total tau, possibly by up-regulating caveolin-1. These actions of rosiglitazone may underlie its neuroprotective effects in the treatment of diffuse axonal injury.
Key Words: nerve regeneration; diffuse axonal injury; rosiglitazone; hyperphosphorylated tau; total tau; caveolin-1; rats; amyloid precursor
protein; ser404; cortex; immunocytochemistry; western blot assay; neural regeneration
and morphological changes in axons (Dong et al., 2014; Lv
There is an urgent need for more effective treatments for
et al., 2014). As a consequence, amyloid-beta precursor pro-
traumatic brain injury, including diffuse axonal injury (DAI).
tein (β-APP) accumulates rapidly and massively, serving as a
In this study, we examined whether rosiglitazone (RSG), a
sensitive biomarker for diagnosis of DAI (Li et al., 2010). Al-
peroxisome proliferator-activated receptor-γ (PPAR-γ) ago-
though there are many promising drug and cell-based ther-
nist, may have therapeutic potential for DAI. DAI initiates a
apeutic approaches for reducing brain injury and enhancing
series of pathophysiological changes including inflammation
functional outcome after DAI, the clinical effectiveness of
and glutamate excitotoxicity, resulting in neurodegeneration,
these approaches is limited (Xiong et al., 2009). Consequent-
neuronal death and neurological dysfunction (Chelly et al.,
ly, further research is needed to explore new therapeutic
2011). During axonal degeneration, hyperphosphorylated
strategies for DAI.
microtubule-associated protein tau dissociates from micro-
RSG is a Food and Drug Administration-approved drug
tubules, leading to total tau loss, microtubule destabilization
with few short-term side effects, and is an excellent candidate
Zhao YL, et al. / Neural Regeneration Research. 2016;11(6):944-950.
for rapid translation to clinical trials. RSG is currently in
bars, a head clip and an anterior teeth hole, with its body at
Phase III clinical trials for Alzheimer's disease, based on its
a 30° angle with respect to the top of the laboratory table.
ability to reduce β-amyloid pathology and inflammation (Liu
When the trigger was pushed, the rat head was rapidly ro-
et al., 2015). RSG has been reported to reduce neuroinflam-
tated 90°, involving sudden acceleration and deceleration.
mation, oxidative stress, apoptotic markers and lesion vol-
All injured rats were in a coma for at least 30 minutes. The
ume in mouse models of traumatic brain injury (Yonutas et
control group only underwent anesthesia and fixation to the
al., 2013; Yao et al., 2015). However, few studies have inves-
device. Rats that died because of injury were excluded and
tigated whether RSG affects tau phosphorylation or tau loss,
later replaced by new rats.
which are pathological features of both Alzheimer's disease and DAI (Xu et al., 2014). Cheng et al. (2014) reported that
RSG suppresses the proliferation of vascular smooth muscle
To ensure that the animal model of DAI used in this study
cells by up-regulating caveolin-1, and that it attenuates cere-
was successful, hematoxylin-eosin staining was performed.
bral vasospasm following experimental subarachnoid hem-
Rats in each group were deeply anesthetized and perfused
orrhage. The up-regulation of caveolin-1 by RSG may also
with 250 mL of normal saline followed by 400 mL of 40 g/L
impact β-APP levels and tau hyperphosphorylation (Hattori paraformaldehyde. The whole brain was removed and post-et al., 2006; Head et al., 2010). However, it remains unknown
fixed in paraformaldehyde. All tissues were desiccated, em-
whether RSG has therapeutic efficacy for DAI. In the present
bedded in paraffin, and sectioned into 5-μm-thick sections.
study, we investigated whether intraperitoneal injection of
Three sections per animal were processed for hematoxy-
RSG rescues the axonal pathology in rats with DAI, and we
lin-eosin staining. Hematoxylin-eosin-stained sections were
examined the underlying mechanisms.
observed at 400× magnification (BX-40; Olympus, Tokyo, Japan).
Materials and Methods
Animals
A total of 108 male 8–10-week-old Sprague-Dawley rats
Brain sections were de-paraffinized in xylene and rehy-
weighing 280–320 g were purchased from the Experimental
drated in a decreasing graded alcohol series and distilled
Animal Center of Xi'an Jiaotong University of China (license
water. Endogenous peroxidase activity was quenched with
No. SCXK (Shaanxi) 2006-001). Animals were housed and
3% H O for 15 minutes, followed by a wash in phos-
fed in a temperature- and humidity-controlled environment
phate-buffered saline (PBS). Sections were placed in 0.01
with a standardized 12-hour reversed light-dark cycle for 1
M citrate buffer and heated in a microwave oven at 95°C
week. This study was carried out in strict accordance with
for 30 minutes. Sections were cooled at room temperature
the recommendations in the Guide for the Care and Use of
for 40 minutes and rinsed in PBS. Non-specific protein
Laboratory Animals of the National Institutes of Health. The
binding was blocked with normal goat serum at room tem-
protocol was approved by the Biomedical Ethics Committee
perature for 30 minutes, followed by incubation with pri-
of Medical College of Xi'an Jiaotong University of China
mary antibodies—rabbit anti-caveolin-1 monoclonal anti-
body (3267P, 1:1,000; Cell Signaling Technology, Danvers, MA, USA), rabbit anti-tau (phospho S404) polyclonal anti-
Establishment of a rat model of DAI and RSG
body (ab131338, 1:200; Abcam, Cambridge, UK) or rabbit
anti-β-APP monoclonal antibody (ab32136, 1:100; Ab-
To investigate changes in levels of β-APP, hyperphosphor-
cam)—for 24 hours at 4°C, followed by a 15-minute wash
ylated tau (phosphorylated at serine 404 (p-tau (S404)) and
in PBS. Sections were then incubated with goat anti-rabbit
total tau after DAI, and to evaluate the effect of RSG, 84 rats
IgG-biotin for 30 minutes (sp9001, 1:200; ZSGB-BIO Co.,
were randomly assigned to 7 groups (n = 12 per group) as
Beijing, China) followed by streptavidin-horseradish per-
follows: control group, DAI 6 hour (h) group, DAI 1 day (d)
oxidase for 30 minutes at 37°C. Sections were washed with
group, DAI 3 d group, DAI 7 d group, DAI 3 d + saline group
PBS for 15 minutes after each step. Diaminobenzidine was
(the same as the DAI 3 d group, but given intraperitoneal
used as the chromogen, and hematoxylin was used as the
injection of saline at 5 minutes, 24 h and 48 h post DAI),
and DAI 3 d + RSG group (the same as the DAI 3 d + saline
Microscopic observation of the immunohistochemical-
group, but administered RSG (Cayman Chemical Co., Ann
ly-stained sections was performed by an experienced pathol-
Arbor, MI, USA), 10 mg/kg intraperitoneal injection, dilut-
ogist blinded to the experimental conditions. Six animals
ed with saline to a final concentration of 2 mg/mL prior to
in each group and five sections per animal were chosen for
injection). Rats were euthanized at the indicated time points
quantitative analysis. Under a light microscope (Olympus),
post injury.
each section was scored in five random visual fields at 400×
The DAI model was established using a lateral head rota-
magnification. Immunoreactivity was scored based on the
tion device, which was created by our team (Li et al., 2013).
number of positive cells and staining intensity using Im-
After anesthesia with 30 g/L pentobarbital sodium (intraper-
age-Pro Plus 6.0 software (Media Cybernetics, Silver Spring,
itoneal injection, 30 mg/kg), the rat's head was horizontally
MD, USA). The immunohistochemical score was obtained
secured to the lateral head rotation device by two lateral ear
by multiplying the staining quantity and intensity scores
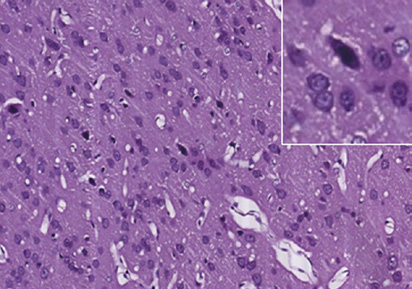
Zhao YL, et al. / Neural Regeneration Research. 2016;11(6):944-950.
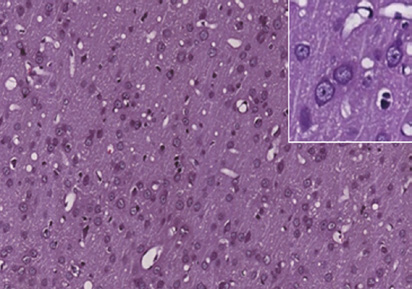
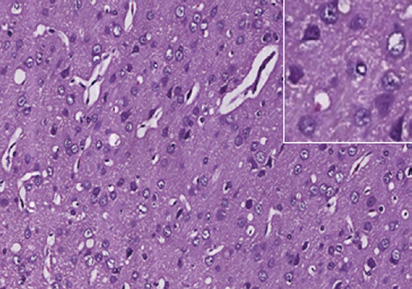
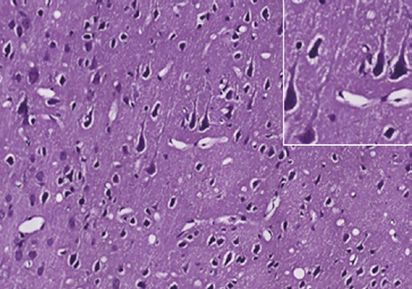
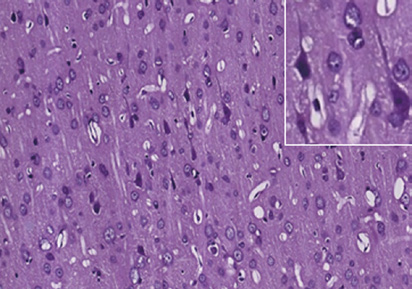
Figure 1 Photomicrographs of the cerebral cortex in the control and DAI groups (hematoxylin-eosin staining).
(A) Control group: no notable abnormality. (B) DAI 6 h group: vacuoles around neurons and pyknosis begin to appear. (C) DAI 1 d group: vacu-
oles are more obvious, and a large number of pyknotic and swollen neurons and distorted axons are observable. (D) DAI 3 d group: the number of
damaged cells appears to be decreased, but pyknotic and swollen neurons and distorted axons are still visible. (E) DAI 7 d group: pyknosis is still
observed, but the tissue appears to be in a recovery stage, scale bars: 100 μm. Figures in the top right corner are magnified images of representative
pathological changes, scale bars: 20 μm. DAI: Diffuse axonal injury; d: day(s); h: hours.
Relative folds of
optical density (β-APP)
Relative folds of
Relative folds of
optical density (p-tau)
optical density (total tau)
Figure 2 Dynamic expression of β-APP, p-tau (S404) and total tau protein in the cortex of control and DAI groups assessed by western blot assay.
Compared with the control group, the levels of β-APP and p-tau (S404) increased at 6 h, peaked at 3 d, and gradually decreased at 7 d after DAI. To-
tal tau declined to the lowest level at 6 h, and then gradually increased after DAI. Histograms show the relative fold changes in each group relative to the control group (mean ± SD, n = 6, one-way analysis of variance followed by Tukey's post hoc test). *P < 0.05, vs. control group. DAI: Diffuse
axonal injury; β-APP: amyloid-beta precursor protein; p-tau (S404): hyperphosphorylated tau at Ser404; d: day(s); h: hours.
(Soslow et al., 2000). The staining quantity was rated on a
20 buffer (TBST) for 2 hours at room temperature, and
scale of 0–4 as follows: 0, no staining; 1, 1–10% cells stained;
incubated overnight with rabbit anti-tau (phospho S404)
2, 11–50%; 3, 51–80%; and 4, 81–100%. Staining intensity
polyclonal antibody (ab131338, 1:1,000; Abcam), rab-
was rated on a scale of 0–3 as follows: 0, negative; 1, weak; 2,
bit anti-β-APP monoclonal antibody (ab32136, 1:1,000;
moderate; and 3, strong. Theoretically, the scores can range
Abcam), mouse anti-tau (tau46) monoclonal antibody
from 0 to 12. An immunohistochemical score of 9–12 was
(4019P, 1:1,000; Cell Signaling Technology) or monoclonal
considered to indicate strong immunoreactivity; 5–8, mod-
rabbit anti-caveolin-1 antibody (3267P, 1:1,000; Cell Sig-
erate; 1–4, weak; and 0, negative.
naling Technology). Membranes were washed three times with TBST for 10 minutes each, and then incubated with
Western blot assay
goat anti-rabbit IgG-horseradish peroxidase secondary
Rats were anesthetized, and the cortex was immediately re-
antibody (ab6721, 1:3,000; Abcam) or goat anti-mouse
moved and stored in liquid nitrogen until processing. Total
IgG-horseradish peroxidase secondary antibody (ab97023,
protein was purified using radioimmunoprecipitation assay
1:3,000; Abcam) for 1 hour at room temperature, with
buffer (Sigma, St. Louis, MO, USA). Protein samples (20
subsequent washing in TBST. β-Actin (ab8227, 1:5,000; Ab-
μg) were analyzed with 10% sodium dodecyl sulfate-poly-
cam, Cambridge, UK) was used as an internal control for
acrylamide gel electrophoresis. Proteins were transferred
protein loading. The membranes were visualized using the
onto polyvinylidene fluoride membranes (Merck Millipore,
ChemiDoc MP System (Bio-Rad, Hercules, CA, USA) with
Darmstadt, Germany). The membranes were blocked with
enhanced chemiluminescence substrate (Millipore). Densi-
5% skimmed milk powder in Tris-buffered saline/Tween
tometric quantification of the bands was performed using
Zhao YL, et al. / Neural Regeneration Research. 2016;11(6):944-950.
Relative folds of optical
density (p-tau) 5
density (total tau)
density (caveolin-1)
Relative folds of optical
Relative folds of optical
Relative folds of optical
Control DAI 3 d DAI 3 d+ DAI 3 d+
Control DAI 3 d DAI 3 d+ DAI 3 d+
Control DAI 3 d DAI 3 d+ DAI 3 d+
Figure 3 Treatment with RSG blocks the increases in β-APP and p-tau (S404) levels and up-regulates the expression of total tau and caveolin-1 in
the cortex 3 d after DAI.
Compared with the control group, the levels of β-APP, p-tau (S404) and caveolin-1 were increased, while total tau was significantly decreased in the
DAI 3 d and DAI 3 d + saline groups. There was no significant difference between the DAI 3 d and DAI 3 d + saline groups. Compared with the DAI 3 d and DAI 3 d + saline groups, the DAI 3 d + RSG group showed increased caveolin-1 and total tau expression and decreased p-tau (S404) and
β-APP levels. Histograms show the relative fold changes in each group relative to the control group (mean ± SD, n = 6, one-way analysis of variance
followed by Tukey's post hoc test). *P < 0.05, vs. the control group; #P < 0.05, vs. the DAI 3 d and DAI 3 d + saline group. DAI: Diffuse axonal inju-
ry; RSG: rosiglitazone; β-APP: amyloid beta-precursor protein; p-tau (S404): hyperphosphorylated tau at Ser404; d: days.
Control DAI 3 d DAI 3 d+saline DAI 3 d+RSG
Scores of IHC (caveolin-1)
Scores of IHC (β-APP)
Scores of IHC (p-tau)
Control DAI 3 d DAI 3 d+ DAI 3 d+
Control DAI 3 d DAI 3 d+ DAI 3 d+
Control DAI 3 d DAI 3 d+ DAI 3 d+
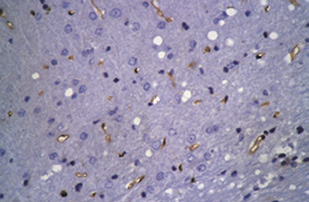
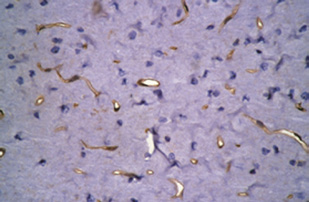
Figure 4 RSG administration inhibits the increases in β-APP and p-tau (S404) and up-regulates caveolin-1 expression in the rat cortex 3 d after
DAI (immunohistochemical staining).
Compared with the control group, strong staining for β-APP and p-tau (S404), and relatively mild staining for caveolin-1 are visible in the cortex of
the DAI 3 d and DAI 3 d + saline groups (*P < 0.05). There was no significant difference between the DAI 3 d and DAI 3 d + saline groups. Com-
pared with the DAI 3 d and DAI 3 d + saline groups, caveolin-1 expression was higher, but p-tau (S404) and β-APP expression was lower in the DAI
3 d + RSG group (#P < 0.05). The histogram shows the immunohistochemical score for each group, determined by multiplying the staining quan-
tity and intensity levels (mean ± SD, n = 6, one-way analysis of variance followed by Tukey's post hoc test). A higher immunohistochemical score indicates stronger staining intensity and/or a greater number of positive cells. Scale bars: 100 μm. DAI: Diffuse axonal injury; RSG: rosiglitazone; IHC: immunohistochemistry; β-APP: amyloid beta-precursor protein; p-tau (S404): hyperphosphorylated tau at Ser404; d: days.
Zhao YL, et al. / Neural Regeneration Research. 2016;11(6):944-950.
Image J software (version 1.29x, NIH, Bethesda, MD, USA).
First, the ratio of the optical density of the protein to that
p-tau (S404) levels were significantly higher in the DAI 3 d
of β-actin was determined. These values in the experimen-
and DAI 3 d + saline groups, with scores of 8.5 and 7.9, re-
tal groups were then normalized to the values in the control
spectively, compared with the control group, which had a
group to obtain relative expression levels.
score of 2.6 (P < 0.05). The DAI 3 d + RSG group showed decreased p-tau (S404) staining with a score of 4.3 (P < 0.05,
vs. DAI 3 d or DAI 3 d + saline groups). Similarly, β-APP
SPSS 17.0 software (SPSS, Chicago, IL, USA) was used for
expression was significantly higher in the DAI 3 d and DAI
statistical analyses. All data were expressed as the mean ± SD.
3 d + saline groups, with scores of 7.6 and 8.2, respectively,
Comparisons among multiple groups were performed using
compared with the control group, which had a score of 1.9 (P
one-way analysis of variance, followed by Tukey's post hoc
< 0.05). β-APP expression in the DAI 3 d + RSG group was
test. A P-value of less than 0.05 was considered statistically
decreased, with a score of 3.6 (P < 0.05, vs. DAI 3 d or DAI 3
d + saline groups). Caveolin-1 was mainly expressed in the cell membrane in the cerebral cortex. Caveolin-1 expression
was significantly higher in the DAI 3 d and DAI 3 d + saline
Histopathological changes in the rat model of DAI
groups, with scores of 3.1 and 3.3, respectively, compared
Hematoxylin-eosin-stained sections showed the presence of
with the control group, which had a score of 1.5 (P < 0.05).
vacuoles around neurons in the cerebral cortex of rats with
RSG treatment increased caveolin-1 expression (score of 6.0;
DAI. Pyknosis was observed in the DAI 6 h, 1 d, 3 d and 7 d
P < 0.05, vs. DAI 3 d or DAI 3 d + saline groups) (Figure 4).
groups (Figure 1B–E). Swollen neurons and distorted axons
were visible in the cerebral cortex of the DAI 6 h and 1 d
groups (Figure 1B, C). Pyknotic, swollen and tangled neu-
Hyperphosphorylation and loss of tau have been implicated
rons were clearly visible in the DAI 1 d group (Figure 1C).
in the pathogenesis of DAI. β-APP accumulates rapidly and
In contrast, in the cortex of rats in the control group, these
massively in axonal bulbs when the axon is damaged (Li et
features were absent (Figure 1A).
al., 2010). Therefore, drugs that simultaneously attenuate tau hyperphosphorylation, lower β-APP levels and inhibit
Dynamic changes in expression of β-APP and in p-tau (S404)
the decrease in total tau may have therapeutic potential in
and total tau levels
the treatment of DAI in patients. In this study, we investi-
Compared with the control group, the expression of β-APP
gated the pathology and dynamic changes in the levels of
was increased 1.6-fold, 2.1-fold, 2.4-fold and 1.9-fold in the
β-APP, p-tau (S404) and total tau at different time points in a
DAI 6 h, 1 d, 3 d and 7 d groups, respectively (P < 0.05).
rat model of DAI. The effects of RSG on β-APP, p-tau (S404),
Compared with the control group, the levels of p-tau (S404)
total tau and caveolin-1 at 3 d after DAI were also assessed.
were increased 6.2-fold, 7.6-fold, 16.7-fold and 7.4-fold, re-
The major findings of our study are as follows: (1) the levels
spectively. Levels of total tau were decreased to 0.5, 0.6, 0.7
of p-tau (S404) and β-APP peaked 3 d post DAI, while total
and 0.9× the levels in the control group in the DAI 6 h, 1 d, 3
tau levels decreased after DAI; (2) RSG treatment attenuated
d and 7 d groups, respectively (all P < 0.05) (Figure 2).
DAI-induced increases in β-APP and p-tau (S404) levels; (3) RSG prevented the decrease in total tau levels 3 d after DAI;
Effects of RSG on β-APP, p-tau (S404), total tau and
(4) RSG treatment increased caveolin-1 expression.
caveolin-1 levels 3 days after DAI
β-APP can be detected at the site of axonal injury. Mu et al.
Western blot assay
(2015) reported that following impact acceleration traumatic
Compared with the control group, β-APP expression was brain injury, β-APP is detectable in injured axons as early as increased 1.6-fold, 1.8-fold and 1.3-fold in the DAI 3 d,
2 to 6 h after trauma and continues to accumulate 1–3 d post
DAI 3 d + saline and DAI 3 d + RSG groups, respectively (all
injury. Hyperphosphorylation of tau results in microtubule
P < 0.05). p-tau (S404) levels were increased 15.1-fold, 16.2-
destabilization. Shultz et al. (2015) showed that in the rat DAI
fold and 7.9-fold in the DAI 3 d, DAI 3 d + saline and DAI
model created by lateral fluid percussion, there is increased
3 d + RSG groups, respectively (all P < 0.05). Caveolin-1
phosphorylation of tau (p-tau (S198/S262)) in the cortex 24 h
expression was increased 1.1-fold, 1.2-fold and 1.6-fold in
and 3 d post injury, although total tau levels did not signifi-
the DAI 3 d, DAI 3 d + saline and DAI 3 d + RSG groups,
cantly differ from the control group (Shultz et al., 2015). In
respectively (all P < 0.05). Total tau levels were decreased to
the present study, β-APP expression and hyperphosphor-
0.4, 0.4 and 0.8× the level in the control group in the DAI 3
ylation of tau, induced by rapid lateral head rotation, had
d, DAI 3 d + saline and DAI 3 d + RSG groups, respectively
similar temporal trends, with significantly increased expres-
(all P < 0.05). There were no significant differences between
sion from 6 h to 3 d, in accordance with the previous study.
the DAI 3 d and DAI 3 d + saline groups (all P > 0.05).
However, p-tau (S404) was detected soon after DAI, while total
Compared with the DAI 3 d or DAI 3 d + saline groups, the
tau was significantly decreased at 6 h, 1 d and 3 d post DAI
DAI 3 d + RSG group showed increased caveolin-1 expres-
in this study. Moreover, the histopathological study revealed
sion and decreased p-tau (S404) and β-APP levels (P < 0.05) the presence of axonal injury 6 h post injury, which greatly
(Figure 3).
worsened 1 to 3 h post injury. These findings suggest that
Zhao YL, et al. / Neural Regeneration Research. 2016;11(6):944-950.
secondary processes contribute to further axonal damage and
tection. Interestingly, the inhibition of PPAR-γ results in a de-
degeneration after the initial injury.
crease in proliferation and loss of the undifferentiated pheno-
RSG is a commonly prescribed insulin-sensitizing drug that
type in neural precursor cells (Bernal et al., 2015). Therefore,
is a selective agonist of PPAR-γ. It has been shown that RSG RSG treatment may also promote nerve regeneration.
provides neuroprotection in animal models of focal ischemia,
In summary, RSG exerts a significant neuroprotective
spinal cord injury, and Alzheimer's disease (Madeira et al.,
effect by suppressing excessive expression of β-APP, by low-
2015). RSG also confers neuroprotection after traumatic brain
ering p-tau (S404) levels, and by preventing the decrease in
injury via anti-inflammatory, anti-apoptotic, anti-oxidative
total tau in a rat model of DAI. Caveolin-1 may be involved
mechanisms (Yi et al., 2008).
in the neuroprotection provided by RSG. Our novel findings
Researches on the effects of RSG on tau hyperphosphor-
suggest that RSG may have therapeutic potential for the
ylation have mainly focused on Alzheimer's disease. RSG
treatment of DAI. Further studies are required to elucidate
alleviates spatial learning deficits in APP/PS1/tau transgenic
the molecular mechanisms underlying the neuroprotective
mice by reducing tau hyperphosphorylation in the brain
effects of RSG.
(Mazanetz and Fischer, 2007; Escribano et al., 2010; Yoon et al., 2010; Tokutake et al., 2012; Song et al., 2014; Yu et al.,
Author contributions: JNS obtained funding, participated
2014). In addition, there are a few reports on the effects of
in the definition of intellectual content of this topic and paper
RSG on β-APP and total tau.
review. YLZ and XDM did literature searching, designed the
The present study is the first to demonstrate that RSG
experiment and collected data. YLZ, XDM, and BFZ wrote
treatment attenuates the dramatic rise in β-APP expression the paper and provided critical revision of the paper. DDL and and p-tau (S404) levels after DAI. Furthermore, RSG inhibited
HGP made the model and contributed to statistical analysis.
the decrease in total tau following DAI, thereby producing a
All authors approved the final version of the paper.
neuroprotective effect. However, the mechanisms underlying
Conflicts of interest: None declared.
these effects of RSG after DAI are still unknown.
Plagiarism check: This paper was screened twice using Cross-
In this study, the down-regulation of β-APP and p-tau Check to verify originality before publication.
(S404) induced by RSG was accompanied by increased ex-
Peer review: This paper was double-blinded and stringently
pression of caveolin-1, a scaffolding protein in caveolae that
reviewed by international expert reviewers.
physically interacts with membrane-associated signaling molecules. RSG dose and time-dependently increases caveo-
lin-1 mRNA and protein levels by activating PPAR-γ and/or Bernal C, Araya C, Palma V, Bronfman M (2015) PPARβ/δ and PPARγ epidermal growth factor receptor (Burgermeister et al., 2003;
maintain undifferentiated phenotypes of mouse adult neural precur-
Llaverias et al., 2004; Seda et al., 2008; Tencer et al., 2008).
sor cells from the subventricular zone. Front Cell Neurosci 9:78-86.
Burgermeister E, Tencer L, Liscovitch M (2003) Peroxisome prolifera-
Up-regulation of caveolin-1 by RSG may mediate some
tor-activated receptor-gamma upregulates caveolin-1 and caveolin-2
of the phenotypic changes in cancer cells, suppress tumor
expression in human carcinoma cells. Oncogene 22:3888-3900.
growth and inhibit proliferation of vascular smooth muscle
Chang CF, Chen SF, Lee TS, Lee HF, Chen SF, Shyue SK (2011) Caveo-
lin-1 deletion reduces early brain injury after experimental intracere-
cells after subarachnoid hemorrhage (Burgermeister et al.,
bral hemorrhage. Am J Pathol 178:1749-1761.
2003; Chintharlapalli et al., 2004; Cheng et al., 2014). When
Chelly H, Chaari A, Daoud E, Dammak H, Medhioub F, Mnif J, Ha-
caveolin-1 is overexpressed, β-APP and β-secretase localize to
mida CB, Bahloul M, Bouaziz M (2011) Diffuse axonal injury in
caveolae, resulting in decreased β-amyloid production, sug-
patients with head injuries: an epidemiologic and prognosis study of 124 cases. J Trauma 71:838-846.
gesting a protective role of caveolin-1 (Hattori et al., 2006).
Cheng MF, Song JN, Li DD, Zhao YL, An JY, Sun P, Luo XH (2014) The
Hyperphosphorylation of tau is observed in hippocampal
role of rosiglitazone in the proliferation of vascular smooth muscle
homogenates from caveolin-1 knockout mice. Knockout of
cells after experimental subarachnoid hemorrhage. Acta Neurochir 156:2103-2109.
caveolin-1 accelerates neurodegeneration and aging (Head et
Chintharlapalli S, Smith R, Samudio I, Zhang W, Safe S (2004) 1, 1-Bis
al., 2010). However, caveolin-1 deletion reduces early brain
(3'-indolyl)-1-(p-substitutedphenyl) methanes induce peroxisome
injury after experimental intracerebral hemorrhage (Chang
proliferator-activated receptor gamma-mediated growth inhibition,
et al., 2011). Our current findings suggest that the up-reg-
transactivation, and differentiation markers in colon cancer cells. Cancer Res 64:5994-6001.
ulation of caveolin-1 produced by RSG is neuroprotective,
Dong DW, Zhang YS, Yang WY, Wang-Qin RQ, Xu AD, Ruan YW (2014)
and may lower β-APP and p-tau (S404) levels and inhibit the
Hyperphosphorylation of tau protein in the ipsilateral thalamus after
reduction in total tau. Further studies are required to clarify
focal cortical infarction in rats. Brain Res 1543:280-289.
the underlying mechanisms of action.
Escribano L, Simon AM, Gimeno E, Cuadrado-Tejedor M, Lopez de
Maturana R, Garcia-Osta A, Ricobaraza A, Perez-Mediavilla A, Del
There are multiple phosphorylation sites on tau, including
Rio J, Frechilla D (2010) Rosiglitazone rescues memory impairment
Ser404, Ser198 and Thr205. The phosphorylation status of these
in Alzheimer's transgenic mice: mechanisms involving a reduced am-
sites differ in different pathological conditions. In this study,
yloid and tau pathology. Neuropsychopharmacology 35:1593-1604.
Hattori C, Asai M, Onishi H, Sasagawa N, Hashimoto Y, Saido TC,
only one site (Ser404) was assessed. Additional phosphoryla-
Maruyama K, Mizutani S, Ishiura S (2006) BACE1 interacts with lip-
tion sites should be examined, and the effects of RSG on neu-
id raft proteins. J Neurosci Res 84:912-917.
rological deficits should also be examined in future studies.
Head BP, Peart JN, Panneerselvam M, Yokoyama T, Pearn ML, Niesman
Furthermore, additional studies are needed to clarify how the
IR, Bonds JA, Schilling JM, Miyanohara A, Headrick J, Ali SS, Roth DM, Patel PM, Patel HH (2010) Loss of caveolin-1 accelerates neuro-
RSG-induced up-regulation of caveolin-1 provides neuropro-
degeneration and aging. PLoS One 5:e15697.
Zhao YL, et al. / Neural Regeneration Research. 2016;11(6):944-950.
Li J, Li XY, Feng DF, Pan DC (2010) Biomarkers associated with diffuse
Tencer L, Burgermeister E, Ebert MP, Liscovitch M (2008) Rosiglitazone
traumatic axonal injury: exploring pathogenesis, early diagnosis, and
induces caveolin-1 by PPARγ-dependent and PPRE-independent
prognosis. J Trauma 69:1610-1618.
mechanisms: the role of EGF receptor signaling and its effect on can-
Li Y, Song J, Liu X, Zhang M, An J, Sun P, Li D, Jin T, Wang J (2013)
cer cell drug resistance. Anticancer Res 28:895-906.
High expression of STIM1 in the early stages of diffuse axonal injury.
Tokutake T, Kasuga K, Yajima R, Sekine Y, Tezuka T, Nishizawa M,
Brain Res 1495:95-102.
Ikeuchi T (2012) Hyperphosphorylation of Tau induced by naturally
Liu J, Wang LN, Jia JP (2015) Peroxisome proliferator-activated re-
secreted amyloid-beta at nanomolar concentrations is modulated
ceptor-gamma agonists for Alzheimer's disease and amnestic mild
by insulin-dependent Akt-GSK3beta signaling pathway. J Biol Chem
cognitive impairment: a systematic review and meta-analysis. Drugs
Aging 32:57-65.
Xiong Y, Mahmood A, Chopp M (2009) Emerging treatments for trau-
Llaverias G, Vazquez-Carrera M, Sanchez RM, Noe V, Ciudad CJ, Lagu-
matic brain injury. Expert Opin Emerg Drugs 14:67-84.
na JC, Alegret M (2004) Rosiglitazone upregulates caveolin-1 expres-
Xu S, Guan Q, Wang C, Wei X, Chen X, Zheng B, An P, Zhang J, Chang L,
sion in THP-1 cells through a PPAR-dependent mechanism. J Lipid
Zhou W, Mody I, Wang Q (2014) Rosiglitazone prevents the memory
Res 45:2015-2024.
deficits induced by amyloid-beta oligomers via inhibition of inflam-
Lv Q, Lan W, Sun W, Ye R, Fan X, Ma M, Yin Q, Jiang Y, Xu G, Dai J,
matory responses. Neurosci Lett 578:7-11.
Guo R, Liu X (2014) Intranasal nerve growth factor attenuates tau
Yao J, Zheng K, Zhang X (2015) Rosiglitazone exerts neuroprotective
phosphorylation in brain after traumatic brain injury in rats. J Neu-
effects via the suppression of neuronal autophagy and apoptosis in
rol Sci 345:48-55.
the cortex following traumatic brain injury. Mol Med Rep 12: 6591-
Madeira JM, Schindler SM, Klegeris A (2015) A new look at auranofin,
dextromethorphan and rosiglitazone for reduction of glia-mediat-
Yi JH, Park SW, Brooks N, Lang BT, Vemuganti R (2008) PPARgamma
ed inflammation in neurodegenerative diseases. Neural Regen Res
agonist rosiglitazone is neuroprotective after traumatic brain injury
via anti-inflammatory and anti-oxidative mechanisms. Brain Res
Mazanetz MP, Fischer PM (2007) Untangling tau hyperphosphoryla-
tion in drug design for neurodegenerative diseases. Nat Rev Drug
Yonutas HM, Sullivan PG (2013) Targeting PPAR isoforms following
Discov 6:464-479.
CNS injury. Curr Drug Targets 14:733-742.
Mu J, Song Y, Zhang J, Lin W, Dong H (2015) Calcium signaling is
Yoon SY, Park JS, Choi JE, Choi JM, Lee WJ, Kim SW, Kim DH (2010)
implicated in the diffuse axonal injury of brain stem. Int J Clin Exp
Rosiglitazone reduces tau phosphorylation via JNK inhibition in the
Pathol 8:4388-4397.
hippocampus of rats with type 2 diabetes and tau transfected SH-
Shultz SR, Wright DK, Zheng P, Stuchbery R, Liu SJ, Sashindranath M,
SY5Y cells. Neurobiol Dis 40:449-455.
Medcalf RL, Johnston LA, Hovens CM, Jones NC, O'Brien TJ (2015)
Yu Y, Li X, Blanchard J, Li Y, Iqbal K, Liu F, Gong CX (2014) Insulin
Sodium selenate reduces hyperphosphorylated tau and improves
sensitizers improve learning and attenuate tau hyperphosphorylation
outcomes after traumatic brain injury. Brain 138:1297-1313.
and neuroinflammation in 3xTg-AD mice. J Neural Transm 122:593-
Seda O, Sedova L, Oliyarnyk O, Kazdova L, Krenova D, Corbeil G,
Hamet P, Tremblay J, Kren V (2008) Pharmacogenomics of metabol-ic effects of rosiglitazone. Pharmacogenomics 9:141-155.
Song JZ, Sun J, Jin DC, Deng YQ (2014) Rosiglitazone improves learn-
ing and memory impairment of 3 x Tg mice. Yao Xue Xue Bao
Copyedited by Patel B, Norman C, Wang J, Qiu Y, Li CH, Song LP, Zhao M
Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, Masferrer
J, Koki AT (2000) COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer 89:2637-2645.
Source: http://www.sjzsyj.org/CN/article/openArticlePDF.jsp?id=1891
International Journal of Systematic and Evolutionary Microbiology (2013), 63, 893–899 Pseudonocardia antitumoralis sp. nov., adeoxynyboquinone-producing actinomyceteisolated from a deep-sea sediment Xin-Peng Tian,1 Li-Juan Long,1 Su-Mei Li,1 Jing Zhang,1 Ying Xu,2Jie He,2 Jie Li,1 Fa-Zuo Wang,1 Wen-Jun Li,2 Chang-Sheng Zhang1and Si Zhang1 1Key Laboratory of Marine Bio-resources Sustainable Utilization, CAS; RNAM Center for Marine
Table of contents Farter, pourquoi donc? Sciolinatura, perché? Testing / Nordic know-how Selbst wer zum ersten Mal auf Skiern steht, eine Même le novice qui, pour la première fois, chausse Persino chi per la prima volta si trova sugli sci, si Loipe betritt oder sich beim Snowboarden versucht – des skis, affronte une piste de ski de fond ou s‘essaie avventura su una pista di fondo o si mette su uno












