
Cialis ist bekannt für seine lange Wirkdauer von bis zu 36 Stunden. Dadurch unterscheidet es sich deutlich von Viagra. Viele Schweizer vergleichen daher Preise und schauen nach Angeboten unter dem Begriff cialis generika schweiz, da Generika erschwinglicher sind.
Ube aromatic sf5 derivatives


Aug, 2012
Aromatic SF5 Derivatives
Prepared in High Yield via Highly Versatile & Cost Competitive Methods
Contact: [email protected]
UBE Aromatic SF5 compounds are expected to be useful as BUILDING
BLOCKS for pharmaceutical agents, pesticides, liquid crystals,
conductive polymer, and higher performance organic materials.
CAS# 1063625-86-4
CAS# 1246998-10-6
CAS# 197384-98-8 CAS# 401892-85-1
CAS# 1126968-75-9
CAS# 1062610-12-1
CAS# 1219501-58-2
CAS# 1219501-60-6
Contact us to inquire about
UBE Industries, Ltd. Pharmacutical Division
Seavans N. Bldg., 1-2-1 Shibaura, Minato-ku, Tokyo 105-8449, Japan
availability and pricing.
Phone: +81-(0)3-5419-6178 FAX: +81-(0)3-5419-6257 E-mail: [email protected]

Aug, 2012
Aromatic hydrocarbon
Phenylsulfur pentafluoride
2-Aminophenylsulfur pentafluoride
CAS# 1246998-10-6: Limited quantities
available for research only
4-Tolylsulfur pentafluoride
3-Aminophenylsulfur pentafluoride
CAS# 203126-21-0
Carboxylic acid and derivatives
4-(Pentafluorosulfanyl)benzoic acid
2-Hydroxyphenylsulfur pentafluoride
4-(Pentafluorosulfanyl)benzoyl chloride
CAS# 1126968-75-9
CAS# 197384-98-8
3-Hydroxyphenylsulfur pentafluoride
CAS# 401892-85-1
4-Hydroxyphenylsulfur pentafluoride
2-Fluorophenylsulfur pentafluoride
CAS# 864230-02-4
3-Fluorophenylsulfur pentafluoride
CAS# 432028-10-9
4-Fluorophenylsulfur pentafluoride
CAS# 1219501-58-2
2,6-Difluorophenylsulfur pentafluoride
CAS# 864230-03-5
2,4,6-Trifluorophenylsulfur pentafluoride
CAS# 1219501-60-6
CAS# 1062610-12-1
Aryl boron reagent
4-Chlorophenylsulfur pentafluoride
3-(Pentafluorosulfanyl)phenylboronic acid
CAS# 871507-67-4
3-Bromophenylsulfur pentafluoride
4-(Pentafluorosulfanyl)phenylboronic acid
CAS# 871507-70-9
4-Bromophenylsulfur pentafluoride


Aug, 2012
UBE Aromatic SF5 derivatives, prepared in high yield via highly
versatile & cost competitive methods.
Introduction: Concurrent with significant developments in the synthetic
methodology for the preparation of SF5 containing compounds, many
potential applications, derived from the interesting and unique properties of
the SF5 function, have been proposed, particularly in certain advanced
specialty chemical fields such as pharmaceuticals, agrochemicals and
electronics.
The SF5 function, one of the most electron-withdrawing groups known,
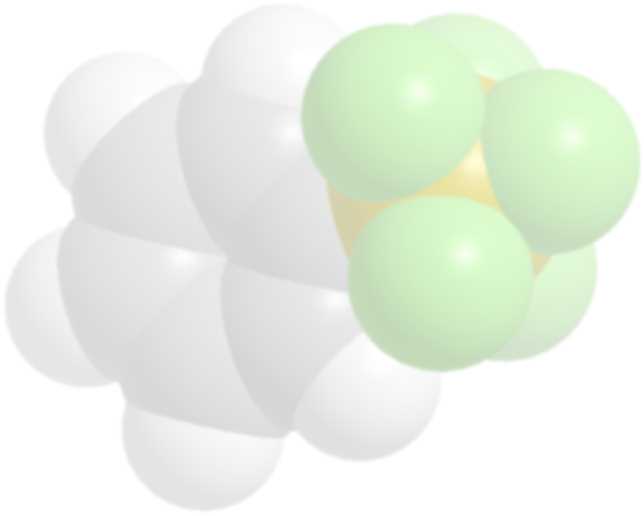
Molecular Model of PhSF5
imparts outstanding lipophilic properties to compounds which incorporate it, as wel as added chemical and thermal stability. It is expected that the higher lipophilicity and other properties of SF5 compounds wil show interesting and unique influences on biological activities other than those observed with fluorine or trifluoromethyl-groups 1).
Regarding electronics chemicals, it is reported that there has been a rapid increase in the number of patents which list the SF5 group and other groups in liquid crystals due to the strong dipole moment which can be achieved by the SF5 group 1), 2).
Properties of Aromatic SF5 compounds:
SF5 group is cal ed "Super-trifluoromethyl group"2), and the expected properties of SF5-containing
compounds are similar to the ones which are seen in general fluorine compounds, although most of them are significantly enhanced by the increment of the number of fluorine atoms in SF5 group.
A) Electron-withdrawing Effect 3)
SF5 group is recognized as a strong electron-withdrawing group. Fig.1 below shows the comparative
values of pKa in the substituted benzoic acid derivatives which have SF5, CF3, SCF3, OCF3 and F,
respectively. In Fig.1, SF5 derivative is ranked as the second strongest group after the nitro-substituted
one.
B) Lipophilicity 2)
It is wel known that compounds which incorporate fluorine(s) show greater lipophilicity. Table 1 shows
the comparative values of lipophilicity with varying substituents in the molecule. SF5 substituted
compounds are expected to show excel ent lipophilicity compared with other fluorine-containing
compounds.
Table 1 Lipophilicity(π) of substituent X
Substituent X SCF3

Aug, 2012
C) Thermal and Chemical Stability
Aromatic SF5 compounds possess excel ent thermal and chemical stability. For example, it was
demonstrated that the thermal decomposition rate of PhSF5 (PSF) was less than 20% after it was heated
in a sealed tube at 400oC for 7 hours 3a). It was also demonstrated that aromatic SF5 compounds are
more tolerant than aromatic CF3 compounds under strong conditions of Brønsted acids and bases 3a, 4a)
and can be widely applied for common synthetic transformations in high yield. Examples of reactions for
Aromatic SF5 compounds are shown below 4).
D) Toxicity
Regarding the assessment of toxicity of Aromatic SF5 compounds, the aromatic SF5 compounds shown
in Table 2 below were assayed for both Ames and Acute Oral Toxicity. Table 2 below shows both results
including the empirical data obtained from the Acute Oral Toxicity test. 4MPSF showed the weak toxicity,
whose range is 50-300mg/Kg and ranked as Category 3 in UN GHS (Table 2).
Table 2 Safety testing of Aromatic SF5 compounds
Ames Test
ACUTE ORAL
TOXICITY (Rat): LD50
E) Biological Activities
Currently, the introduction of fluorine into organic molecules has become very common methodology in
biomedical fields, and numerous fluorine containing molecules have been developed and many have
shown significant promise and advantages in this field.
In particular, the pentafluorosulfanyl (SF5) group, which is a highly fluorinated functional group, has shown remarkable activity in biochemical molecules. The introduction of the SF5 group brings not only the

Aug, 2012
novel properties which originate from Fluorine element (Strong electronegativity, high lipophilicity and high chemical stability) to the molecule, but also a larger steric effect than the CF3 group, which is also recognized as a highly fluorinated functional group. The relative steric demand of the SF5 group is slightly less than that of a tert-butyl group and considerably larger than that of a CF3 group 5). Examples of biological activities comparing the CF3 substituted agent vs. the SF5 analog are shown below;
Mefloquine is used for both treatment and prophylaxis of malaria (Fig.
2). 8-SF5-Mefloquine showed a longer half-life(68h) than
Mefloquine(23h) after administration to mice 4e), 6). Fipronil is a broad
spectrum insecticide (Table 3).SF5 analogue of Fipronil was not only
more active than Fipronil but showed no loss of potency towards the
resistant strain of housefly, in contrast to the Fipronil 7).
Table 3. Comparison of the insecticidal activity
a) (R) indicates a strain resistant to dieldrin and (S) is a susceptible strain.
SF5 Analogue
UBE Preparation Methods: In order to contribute to and improve SF5 chemistry above, UBE has started
to deliver a series of aromatic pentafluorosulfanyl compounds prepared by new innovative processes
including our KF/Cl2 method, which was developed by IM&T Research Inc. (Scheme 1) 8).
Our KF/Cl2 method is widely applicable to various aromatic disulfide compounds, which are direct starting materials for the corresponding aromatic SF5 compounds. This has enabled us to introduce the SF5 group into various aromatic rings via a 2 step process from the corresponding aryl-disulfide, as compared to the direct fluorination process utilizing elemental fluorine, which is limited by the use of only nitro-aryl compounds as starting materials 9).
With the KF/Cl2 process, aryl-disulfide is converted to the corresponding aryl tetrafluorosulfanyl- chloride. This process is equal y applicable to aromatic thiophenol compounds. The obtained Aryl-SF4Cl from the KF/Cl2 process can then be converted to the corresponding aryl-pentafluoro- sulfanyl compound with zinc difluoride or anhydrous HF. Aryl-SF4Cl preparation proceeds with high yield around 80-90% at room temperature, and the starting materials provided for this reaction, aryl-disulfide, KF and Cl2, are commodity materials, which can be obtained conveniently and at relatively low prices for industrial scale production.
The conversion to Aryl-SF5 from the corresponding Aryl-SF4Cl, proceeds with high yield (around 70-80%) with zinc difluoride at 100°C, and it also has been demonstrated that this reaction proceeds with aHF in high yield (70-75%) below 20°C.
Aug, 2012
Products: Based on these patented synthetic technologies 10), UBE has prepared the fol owing Aryl-SF5
compounds in page 1, which can be employed as "building blocks" for the preparation of more valuable SF5
compounds by customers. To elaborate on our highly extensive and applicable methodology, we have
succeeded in applying it to the preparation of aromatic poly-SF5 compounds. Also, UBE will continue to
develop and produce additional novel new Aryl-SF5 compounds. Some new compounds are il ustrated in Fig.
3.
New!
CAS# 1126968-75-9
Contact: For quotes, requests, and other questions, please contact us directly at the address below;
Ube Industries, Ltd., Pharmacutical Division
Seavans N. Bldg., 1-2-1 Shibaura, Minato-ku, Tokyo 105-8449, Japan
Phone: +81-(0)3-5419-6178 FAX: +81-(0)3-5419-6257
E-mail: [email protected]
References:
R. W. Winter, R. A. Dodean, and G. L.Gard, Fluorine-Containing Synthons, V. A. Soloshonok edited; American
Chemical Society, Washington,2005, 911, 87.
P. Kirsch, Modern Fluoroorganic Chemistry; WILEY-VCH, Weinheim, 2004,146.
a) W. A. Sheppard, J. Am. Chem. Soc. 1962, 84, 3072-76. b) C. J. Byrne, et al., J. Chem. Soc. Perkin Trans. 2 1987,
1649-53. c) J. Shorter, Pure Appl. Chem. 1997, 69, 2497-2510.
a) R. D. Bowden et al., Tetrahedron 2000, 56, 3399-3408. b) P. Kirsch et al., Angew. Chem. Int. Ed. 1999, 38,
1989-1992. c) S. Nishino et al., JP Patent 2009-96740, 2009. d) A. M. Sipyagin et al., J. Fluorine Chem. 2004, 125,
1305-1316. e) T. Mo et al., Tetrahedron Lett., 2010, 51, 5137-5140.
D. Lentz et al., In Chemistry of Hypervalent Compounds, K. Akiba, (Eds), Wiley-VCH, New York, 1998, 295.
G. S. Dow et al., WO 2010/144434, 2010.
P. J. Crowley et al., CHIMIA 2004, 58, 138-142.
T. Umemoto et al., Beilstein J. Org. Chem. 2012, 8, 461–471.
R. D. Bowden et al., WO 97/05106, 1997.
10) T. Umemoto et al., US 7592491, 2009.
Source: https://www.ube.com/products/pharmaceutical/sf5-chemicais.pdf
Latin American Pharmacogenomics and Personalized MedicineConference 2nd Latin American Pharmacogenomics and Personalized Medicine Congress27–29 June 2012, Rio de Janeiro, RJ, Brazil There are nearly 600 million people living in 24 Latin American countries, speaking two major languages (Portuguese and Spanish) and sharing ancestral roots in America, Europe and Africa. Ethnic and cultural diversity, socioeconomical, scientific and technological disparities across Latin America must be taken into account in the design, interpretation and implications of pharmacogenomic studies in this region. The conference covered some of these aspects, but also took on a more global approach on the growing contribution of genomic information and biotechnological tools to the way medicines are developed, regulated and prescribed to patients. Translation of pharmacogenomics into clinical practice was the topic of a keynote lecture and two debate sessions. A preconference Introductory Course of Pharmacogenomics was offered.
British Journal of Pharmacology (2001) 132, 879 ± 888 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 A1 adenosine receptors inhibit multiple voltage-gated Ca2+ channel subtypes in acutely isolated rat basolateral amygdala neurons*,1Brian A. McCool & 1Jeery S. Farroni 1Department of Medical Pharmacology and Toxicology, The Texas A&M University System Health Science Center, College












