
Cialis ist bekannt für seine lange Wirkdauer von bis zu 36 Stunden. Dadurch unterscheidet es sich deutlich von Viagra. Viele Schweizer vergleichen daher Preise und schauen nach Angeboten unter dem Begriff cialis generika schweiz, da Generika erschwinglicher sind.
Pain_brochure_2011.qxp
A Note from Americans for Safe Access
We are committed to ensuring safe, legal availability of marijuana formedical uses. This brochure is intended to help doctors, patients andpolicymakers better understand how marijuana—or "cannabis" as it ismore properly called—may be used as a treatment for people with seri-ous medical conditions. This booklet contains information about usingcannabis as medicine. In it you'll find information on:
Why Cannabis is Legal to Recommend . . . . . . . . . . . . .3Overview of the Scientific Research on Medical Cannabis . . . . 4Research on Cannabis and Chronic Pain . . . . . . . . . . . . 6Comparison of Medications: Efficacy and Side-Effects . . . . . .9 Why Cannabis is Safe to Recommend . . . . . . . . . . . . .11Testimonials of Patients and Doctors . . . . . . . . . . . . .13History of Cannabis as Medicine . . . . . . . . . . . . . . . 19Scientific and Legal References . . . . . . . . . . . . . . . .22
We recognize that information about using cannabis as medicine hasbeen difficult to obtain. The federal prohibition on cannabis has meantthat modern clinical research has been limited, to the detriment ofmedical science and the wellness of patients. But the documented histo-ry of the safe, medical use of cannabis dates to 2700 B.C. Cannabis waspart of the American pharmacopoeia until 1942 and is currently avail-able by prescription in the Netherlands and Canada.
Testimonials from both doctors and patients reveal valuable informa-tion on the use of cannabis therapies, and supporting statements fromprofessional health organizations and leading medical journals supportits legitimacy as a medicine. In the last few years, clinical trials in GreatBritain, Canada, Spain, Israel, and elsewhere have shown great promisefor new medical applications.
This brochure is intended to be a starting point for the consideration ofapplying cannabis therapies to specific conditions; it is not intended toreplace the training and expertise of physicians with regard to medi-cine, or attorneys with regard to the law. But as patients, doctors andadvocates who have been working intimately with these issues formany years, Americans for Safe Access has seen firsthand how helpfulcannabis can be for a wide variety of indications. We know doctorswant the freedom to practice medicine and patients the freedom tomake decisions about their healthcare.
For more information about ASA and the work we do, please see ourwebsite at AmericansForSafeAccess.org or call 1-888-929-4367.
Americans for Safe Access
Is Cannabis Legal to Recommend?
In 2004, the United States Supreme Court upheld earlier federal courtdecisions that doctors have a fundamental Constitutional right to rec-ommend cannabis to their patients.
The history. Within weeks of California voters legalizing medicalcannabis in 1996, federal officials had threatened to revoke the pre-scribing privileges of any physicians who recommended cannabis totheir patients for medical use.1 In response, a group of doctors andpatients led by AIDS specialist Dr. Marcus Conant filed suit against thegovernment, contending that such a policy violates the First Amend-ment.2 The federal courts agreed at first the district level,3 then all theway through appeals to the Ninth Circuit and then the Supreme Court.
What doctors may and may not do. In Conant v. Walters,4 the NinthCircuit Court of Appeals held that the federal government could nei-ther punish nor threaten a doctor merely forrecommending the use of cannabis to apatient.5 But it remains illegal for a doctor to"aid and abet" a patient in obtainingcannabis.6 This means a physician may discussthe pros and cons of medical cannabis withany patient, and issue a written or oral rec-ommendation to use cannabis without fearof legal reprisal.7 This is true regardless ofwhether the physician anticipates that thepatient will, in turn, use this recommenda-tion to obtain cannabis.8
What physicians may not do is actually pre-scribe or dispense cannabis to a patient9 or
Angel Raich & Dr. Frank Lucido
tell patients how to use a written recommendation to procure it from acannabis club or dispensary.10 Doctors can tell patients they may behelped by cannabis. They can put that in writing. They just can't helppatients obtain the cannabis itself.
Patients protected under state, not federal, law. In June 2005, the U.S.
Supreme Court overturned the Raich v. Ashcroft Ninth Circuit Court ofAppeals decision. In reversing the lower court's ruling, Gonzales v. Raichestablished that it is legal under federal law to prosecute patients whopossess, grow, or consume medical cannabis in medical cannabis states.
However, this Supreme Court decision does not overturn or supersedethe laws in states with medical cannabis programs.
For assistance with determining how best to write a legal recommenda-tion for cannabis, please contact ASA at 1-888-929-4367.
Scientific Research Supports Medical Cannabis
Between 1840 and 1900, European and American medical journals pub-lished more than 100 articles on the therapeutic use of the drug knownthen as Cannabis Indica (or Indian hemp) and now simply as cannabis.
Today, studies published in peer-reviewed journals demonstrate cannabishas medical value in treating patients with serious illnesses such as AIDS,glaucoma, cancer, multiple sclerosis, epilepsy, arthritis, and chronic pain.
The safety of the drug has been attested to by numerous studies andreports, including the LaGuardia Report of 1944, the Schafer CommissionReport of 1972, a 1997 study conducted by the British House of Lords, theInstitutes of Medicine report of 1999, research sponsored by Health Canada,and numerous studies conducted in the Netherlands, where cannabis has
been quasi-legal since 1976 and iscurrently available from pharma-
INSTITUTE OF MEDICINE
cies by prescription.
"Nausea, appetite loss, pain and anxiety
Recent published research on CD4
. all can be mitigated by marijuana.
immunity in AIDS patients found
For patients, such as those with AIDS or
no compromise to the immune
undergoing chemotherapy, who suffer
systems of patients undergoing
simultaneously from severe pain, nau-
cannabis therapy in clinical trials.11
sea, and appetite loss, cannabinoid drugsmight offer broad spectrum relief not
The use of medical cannabis has
found in any other single medication."
been endorsed by numerous pro-fessional organizations, including
Marijuana and Medicine:
the American Academy of Family
Assessing the Science Base, 1999
Physicians, the American PublicHealth Association, and the
American Nurses Association. Its use is supported by such leading medicalpublications as The New England Journal of Medicine and The Lancet.
Recent Research Advances
While research has until recently been sharply limited by federal prohibi-tion, the last few years have seen rapid change. More than 15,000 modernpeer-reviewed scientific articles on the chemistry and pharmacology ofcannabis and cannabinoids have been published, as well as more than 2,000articles on the body's natural endocannabinoids. The InternationalCannabinoid Research Society was formally incorporated as a scientificresearch organization in 1991. Membership in the Society has more thantripled from about 50 members in the first year to over 500 in 2010. TheInternational Association for Cannabis as Medicine (IACM) was founded inMarch 2000. It publishes a bi-weekly newsletter and the IACM-Bulletin, andholds a bi-annual symposium to highlight emerging research in cannabistherapeutics.
Americans for Safe Access
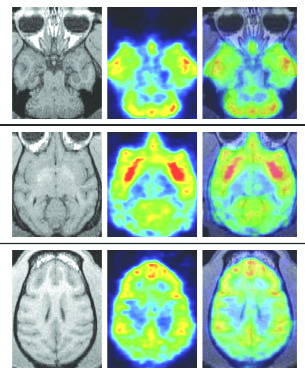
In 2001, the State of California established the Center for MedicinalCannabis Research to coordinate an $8.7-million research effort at Universityof California campuses. As of 2010, the CMCR had completed six of 14approved studies. Of those, five published double-blind, placebo-controlledstudies studied pain relief; each showed cannabis to be effective. A 2009review of controlled clinical studies conducted over a 38-year period, foundthat "nearly all of the 33 publishedcontrolled clinical trials conducted inthe United States have shown signifi-cant and measurable benefits in sub-jects receiving the treatment."12 Thereview's authors note that the morethan 100 different cannabinoids incannabis have the capacity for analge-sia through neuromodulation inascending and descending pain path-ways, neuroprotection, and anti-inflammatory mechanisms.
In the United Kingdom, GWPharmaceuticals has been conductingclinical trials with its cannabis-basedmedicine, Sativex®, for the pastdecade. GW's Phase II and Phase III tri-als of cannabis-based medicine showpositive results for the relief of neuro-
Cannabinoid receptors in the brain
logical pain related to: multiple sclero-sis (MS), spinal cord injury, peripheral nerve injury (including peripheral neu-ropathy secondary to diabetes mellitus or AIDS), central nervous systemdamage, neuroinvasive cancer, dystonias, cerebral vascular accident, andspina bifida. They have also shown cannabinoids to be effective in clinicaltrials for the relief of pain and inflammation in rheumatoid arthritis andalso pain relief in brachial plexus injury.13-16
As of December 2010, the company has obtained regulatory approval inSpain, New Zealand, and the UK for Sativex® Oromucosal Spray, a con-trolled-dose whole-plant extract. Sativex® was approved in Canada forsymptomatic relief of neuropathic pain in 2005, in 2007 for patients withadvanced cancer whose pain is not fully alleviated by opiods, and in 2010for spasticity related to multiple sclerosis. Sativex® has been made availableeither for named patient prescription use or for clinical trials purposes in atotal of 22 countries. In the US, GW was granted an import license forSativex® by the DEA following meetings in 2005 with the FDA, DEA, theOffice for National Drug Control Policy, and the National Institute for DrugAbuse. Sativex® is currently an investigational drug in FDA-approved clinicaltrials as an adjunctive analgesic treatment for patients with advanced cancerwhose pain is not relieved by strong opioids.
CANNABIS AND CHRONIC PAIN
Persistent and disabling pain can have numerous and sometimes multiplecauses. Among them are cancer; AIDS; sickle cell anemia; multiple sclerosis;defects or injuries to the back, neck and spinal cord; arthritis and otherrheumatic and degenerative hip, joint and connective tissue disorders; andsevere burns. Pain is not a primary condition or injury, but rather a severe,frequently intolerable symptom that varies in frequency, duration, and
severity according to the individual. Theunderlying condition determines the appro-priate curative approach, but does notdetermine the proper symptom manage-ment. It is the character, severity, locationand duration of the pain that determinesthe range of appropriate therapies.
Chronic pain is a public health issue that iswidespread across the aging populations ofindustrialized nations. Epidemiological sta-tistics are alarming: In Europe, it is estimat-
ed that one in four adults has a chronic pain condition.17 In the US, it is esti-mated that at least 38 million adults suffer from chronic pain, and at least12 million have used cannabis as a treatment.
For patients in pain, the goal is to function as fully as possible by reducingtheir pain as much as possible, while minimizing the often-debilitating sideeffects of the pain therapies. Failure to adequately treat severe and/orchronic pain can have tragic consequences. Not infrequently, people in unre-lieved pain want to die. Despair can also cause patients to discontinuepotentially life-saving procedures (e.g., chemotherapy or surgery), whichthemselves cause severe suffering. In such dire cases, anything that helps toalleviate the pain will prolong these patients' lives.
Cannabis can serve at least two important roles in safe, effective pain man-agement. It can provide relief from the pain itself (either alone or in combi-nation with other analgesics), and it can control the nausea associated withtaking opioid drugs, as well as the nausea, vomiting and dizziness thatoften accompany severe, prolonged pain.
Opioid therapy is often an effective treatment for severe pain, but all opi-ates have the potential to induce nausea. The intensity and duration of thisnausea can cause enormous discomfort and additional suffering and lead tomalnourishment, anorexia, wasting, and a severe decline in a patient'shealth. Some patients find the nausea so intolerable that they are inclinedto discontinue the primary pain treatment, rather than endure the nausea.
Inhaled cannabis provides almost immediate relief for nausea with signifi-cantly fewer adverse side effects than orally ingested Marinol. Inhalation
Americans for Safe Access
allows the active compounds in cannabis to be absorbed into the bloodstream with greater speed and efficiency. It is for this reason that inhalationis an increasingly common, and often preferable, route of administrationfor many medications. Cannabis may also be more effective than Marinolbecause it contains many more cannabinoids than just the THC that isMarinol's active ingredient. The additional cannabinoids may well haveadditional and complementary antiemetic qualities. They have been conclu-sively shown to have better pain-control properties when taken in combina-tion than THC alone, and mitigate anxiety and other side-effects of THC.
Research on cannabis and pain management
Cannabis has been used as an analgesic for thousands of years18-20 andpatients often report significant pain relief from cannabis, even in caseswhere conventional pain therapies have failed.21-26
After reviewing a series of trials in 1997, the U.S. Society for Neuroscienceconcluded that "substances similar to or derived from marijuana could ben-efit the more than 97 million Americanswho experience some form of pain eachyear."27 A 1999 study commissioned by theWhite House and conducted by theInstitute of Medicine recognized the rolethat cannabis can play in treating chronicpain.28 "After nausea and vomiting, chron-ic pain was the condition cited most oftento the IOM study team as a medicinal usefor marijuana."
From 1975 to February 2011, there havebeen nearly 300 studies showing thatcannabinoids and cannabis can help
patients experiencing chronic pain.29,30
Some of the most encouraging clinical data on effects of cannabinoidson chronic pain are from studies of intractable cancer pain and hard-to-treat neuropathic pain.31 The effectiveness of cannabis and cannabi-noids in relieving neuropathic pain has been demonstrated in morethan three dozen preclinical and clinical trials.32 A trial of cannabis ciga-rettes to treat HIV-associated daily neuropathic pain in 50 patients showedan average reduction of pain by 30% over a treatment course of only 5days.33 In 2001, researchers reported that cannabis extract sprayed under thetongue (Sativex®) was effective in reducing pain in patients sufferingintractable neuropathic pain.34 A review of over 20 clinical trials on cannabisand cannabinoids found that whole plant cannabis and extracts are superiorto oral THC for the treatment of pain. Health Canada approved Sativex® forprescription in the treatment of HIV-associated neuropathic pain in 2005and cancer pain in 2007.
The activity of the more than 100 cannabinoids and other components onthe plant may explain its superiority in reducing pain when comparingwhole plant cannabis and extracts to THC alone. For instance, the cannabi-noid cannabichromene (CBC), the third most common ingredient on the
plant, exhibits anti-inflamma-tory and analgesic actions,
FEDERATION OF AMERICAN SCIENTISTS
although weaker than THC.35
"Based on much evidence, from patients and
Similarly, beta-sitosterol, a
doctors alike, on the superior effectiveness
and safety of whole cannabis compared to
found in cannabis, was able
other medications,… the President should
to decrease inflammation and
instruct the NIH and the FDA to make efforts
edema in skin treatment.36
to enroll seriously ill patients whose physi-
And a unique flavanoid found
cians believe that whole cannabis would be
only in cannabis, cannaflavin
helpful to their conditions in clinical trials"
A, inhibits the inflammatorymolecule PGE-2, thirty times
FAS Petition on Medical Marijuana, 1994
more potently than aspirin.37Lastly beta-caryophyllene, a
cannabinoid found in many plants besides cannabis, has strong anti-inflam-matory properties but no noticeable side effects.38 Beta-caryophyllen is themost commonly consumed FDA-approved cannabinoid in food.
The IOM report found that "basic biology indicates a role for cannabinoidsin pain and control of movement, which is consistent with a possible thera-peutic role in these areas. The evidence is relatively strong for the treatmentof pain and intriguingly, although less well established, for movement disor-der." According to the IOM Report and numerous independent researcharticles, a number of areas in the brain that have an established role in sens-ing and processing pain respond to the analgesic effect of cannabis, addingthat cannabinoids have been used successfully to treat cancer pain, which isoften resistant to treatment with opiates. The effectiveness of cannabinoidsin treating intractable cancer pain has been demonstrated in several subse-quent clinical trials of a dosage-controlled sublingual spray.
Several studies have found that cannabinoids have analgesic effects in ani-mal models, sometimes equivalent to codeine.39-43 Cannabinoids also seem tosynergize with opioids, which often lose their effectiveness as patients buildup tolerance. One study found morphine was 15 times more active in ratswith the addition of a small dose of THC. Codeine was enhanced on theorder of 900 fold.44 In 1990, researchers conducted a double-blind studycomparing the antispasmodic and analgesic effects of THC, oral Codeine,and a placebo on a single patient suffering from a spinal cord injury.45 Theirfindings confirmed the analgesic effects of THC being "equivalent tocodeine." A 1997 study made similar findings related to morphine.46
A 1999 article reviewing the body of scientific animal research concerningthe analgesic effects of marijuana concludes that "[t]here is now unequivo-
Americans for Safe Access
cal evidence that cannabinoids are antinociceptive [capable of blocking theappreciation or transmission of pain] in animal models of acute pain."47
The report further notes that multiple cannabinoids and noncannabinoidcomponents can serve as anti-inflammatory agents, and so have potential inpreventing and reducing pain caused by swelling (such as arthritis).
In short, the research community recognizes the potential benefits ofcannabis for certain patients, including:
• Chemotherapy patients, especially those being treated for mucositis,
nausea, and anorexia.
• Postoperative pain patients (using cannabinoids as an opioid adjunct to
reduce the nausea and vomiting).
• Patients with spinal cord injury, peripheral neuropathic pain, or central
post-stroke pain.
• Patients with chronic pain and insomnia.
• AIDS patients with cachexia, AIDS neuropathy, or any significant pain.
Britain's House of Lords reached similar conclusions and called for makingcannabis available by prescription.48
How cannabis compares to other medications
According to the Institute of Medicine, "All of the currently available anal-gesic (pain-relieving) drugs have limited efficacy for some types of pain.
Some are limited by dose-related side effects and some by the developmentof tolerance or dependence."
The opioid analgesics commonly used to combat pain include codeine(Dolacet, Hydrocet, Lorcet, Lortab, Vicodin); morphine (Avinza, Oramorph);oxycodone (Oxycontin, Roxicodone, Percocet, Roxicet); propoxyphene(Darvon, Darvocet) and tramadol (Ultram, Ultracet). These medicines cancause psychological and physical dependence, as well as constipation, dizzi-ness, lightheadedness, mood changes, nausea, sedation, shortness of breathand vomiting. Taking high doses or mixing with alcohol can slow downbreathing, a potentially fatal condition.
In addition, patients in pain are often prescribed muscle relaxants such asRobaxin and Flexeril; anti-anxiety agents like Valium, Sinequan, Vistaril,Ativan and Xanax; hypnotics such as Halcion, Restoril, Chloralhydrate,Dalmane and Doral and antiemetics like Zofran, Compazine, Phenergan,Tigan and Marinol.
Robaxin's side effects include abnormal taste, amnesia, blurred vision, confu-sion, dizziness, drop in blood pressure and fainting, drowsiness, fever, flushing,headache, hives, indigestion, insomnia, itching, light-headedness, nasal conges-tion, nausea, pinkeye, poor coordination, rash, seizures, slowed heartbeat,uncontrolled eye movement, vertigo, vomiting and yellow eyes and skin.
Flexeril can cause abnormal heartbeats, aggressive behavior, agitation, anxi-ety, bloated feeling, blurred vision, confusion, constipation, convulsions,decreased appetite, depressed mood, diarrhea, difficulty falling or stayingasleep, difficulty speaking, disorientation, double vision, excitement, faint-ing, fatigue, fluid retention, gas, hallucinations, headache, heartburn, hepa-titis, hives, increased heart rate, indigestion, inflammation of the stomach,itching, lack of coordination, liver diseases, loss of sense of taste, low bloodpressure, muscle twitching, nausea, nervousness, palpitations, paranoia,rash, ringing in the ears, severe allergic reaction, stomach and intestinalpain, sweating, swelling of the tongue or face, thirst, tingling in hands orfeet, tremors, unpleasant taste in the mouth, urinating more or less thanusual, vague feeling of bodily discomfort, vertigo, vomiting, weakness, andyellow eyes and skin.
The newer antiemetics, Anzamet, Kytril and Zofran, are serotonin antago-nists, blocking the neurotransmitter that sends a vomiting signal to thebrain. Rare side effects of these drugs include fever, fatigue, bone pain,muscle aches, constipation, loss of appetite, inflammation of the pancreas,changes in electrical activity of heart, vivid dreams, sleep problems, confu-sion, anxiety and facial swelling.
Reglan, a substituted benzamide, increases emptying of the stomach, thusdecreasing the chance of developing nausea and vomiting due to foodremaining in the stomach. When given at high doses, it blocks the messagesto the part of the brain responsible for nausea and vomiting. Side effectsinclude sleepiness, restlessness, diarrhea and dry mouth. Rarer side effectsare rash, hives and decreased blood pressure
Haldol and Inapsine are tranquilizers that block messages to the part of thebrain responsible for nausea and vomiting. Possible side effects includedecreased breathing rate, increased heart rate, decrease in blood pressurewhen changing position and, rarely, change in electrical activity of theheart.
Compazine and Torecan are phenothiazines, the first major anti-nauseadrugs. Both have tranquilizing effects. Common side effects include drymouth and constipation. Less common effects are blurred vision, restless-ness, involuntary muscle movements, tremors, increased appetite, weightgain, increased heart rate and changes in electrical activity of heart. Rareside effects include jaundice, rash, hives and increased sensitivity to sunlight.
Benadryl, an antihistamine, is given along with Reglan, Haldol, Inapsine,Compazine and Torecan to counter side effects of restlessness, tongue pro-trusion, and involuntary movements. Its side effects include sedation,drowsiness, dry mouth, dizziness, confusion, excitability and decreasedblood pressure.
Benzodiazepine drugs Ativan and Xanax are prescribed to combat the anxi-
Americans for Safe Access
ety associated with chronic pain. Ativan causes amnesia. Abruptly stoppingthe drug can cause anxiety, dizziness, nausea and vomiting, and tiredness. Itcan cause drowsiness, confusion, weakness, and headache when first start-ing the drug. Nausea, vomit-ing, dry mouth, changes inheart rate and blood pres-
AMERICAN ACADEMY OF FAMILY PHYSICIANS
sure, and palpitations are
"The American Academy of Family Physicians
possible side effects.
[supports] the use of marijuana . under med-ical supervision and control for specific med-
Cannabis: By comparison,
ical indications."
the side effects associatedwith cannabis are typically
1996-1997 AAFP Reference Manual
mild and are classified as"low risk." Euphoric moodchanges are among the most frequent side effects. Cannabinoids can exac-erbate schizophrenic psychosis in predisposed persons. Cannabinoidsimpede cognitive and psychomotor performance, resulting in temporaryimpairment. Chronic use can lead to the development of tolerance.
Tachycardia and hypotension are frequently documented as adverse eventsin the cardiovascular system. A few cases of myocardial ischemia have beenreported in young and previously healthy patients. Inhaling the smoke ofcannabis cigarettes induces side effects on the respiratory system.
Cannabinoids are contraindicated for patients with a history of cardiacischemias. In summary, a low risk profile is evident from the literature avail-able. Serious complications are very rare and are not usually reported dur-ing the use of cannabinoids for medical indications.
Is cannabis safe to recommend?
"The smoking of cannabis, even long term, is not harmful to health." Sobegan a 1995 editorial statement of Great Britain's leading medical journal,The Lancet. The long history of human use of cannabis also attests to itssafety—nearly 5,000 years of documented use without a single death. Inthe same year as the Lancet editorial, Dr. Lester Grinspoon, a professoremeritus at Harvard Medical School who has published many influentialbooks and articles on medical use of cannabis, had this to say in an article inthe Journal of the American Medical Association (1995):
"One of marihuana's greatest advantages as a medicine is its remark-able safety. It has little effect on major physiological functions. There isno known case of a lethal overdose; on the basis of animal models, theratio of lethal to effective dose is estimated as 40,000 to 1. By compari-son, the ratio is between 3 and 50 to 1 for secobarbital and between 4and 10 to 1 for ethanol. Marihuana is also far less addictive and far lesssubject to abuse than many drugs now used as muscle relaxants, hyp-notics, and analgesics. The chief legitimate concern is the effect ofsmoking on the lungs. Cannabis smoke carries even more tars and
other particulate matter than tobacco smoke. But the amount smokedis much less, especially in medical use, and once marihuana is an open-ly recognized medicine, solutions may be found; ultimately a technolo-gy for the inhalation of cannabinoid vapors could be developed."49
The technology Dr. Grinspoon imagined in 1995 now exists in the form of"vaporizers," (which are widely available through stores and by mail-order)and recent research attests to their efficacy and safety.50 Additionally, phar-maceutical companies have developed sublingual sprays and tablet forms ofthe drug. Patients and doctors have found other ways to avoid the potential
problems associated with smok-ing, though long-term studies ofeven the heaviest users inJamaica, Turkey and the U.S.
have not found increased inci-dence of lung disease or otherrespiratory problems.
A decade-long study of 65,000Kaiser-Permanente patients com-paring cancer rates among non-smokers, tobacco smokers, andcannabis smokers found thatthose who used only cannabis
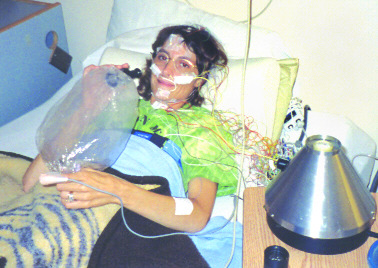
Angel Raich using a vaporizer
had a slightly lower risk of lungand other cancers as compared
to non-smokers.51 Similarly, a study comparing 1,200 patients with lung,head and neck cancers to a matched group with no cancer found that eventhose cannabis smokers who had consumed in excess of 20,000 joints hadno increased risk of cancer.52
As Dr. Grinspoon notes, "the greatest danger in medical use of marihuana isits illegality, which imposes much anxiety and expense on suffering people,forces them to bargain with illicit drug dealers, and exposes them to thethreat of criminal prosecution." This was the same conclusion reached bythe House of Lords report, which recommended rescheduling and decrimi-nalization.
Cannabis or Marinol?
Those committed to the prohibition on cannabis frequently cite Marinol, aSchedule III drug, as the legal means to obtain the benefits of cannabis.
However, Marinol, which is a synthetic form of THC, does not deliver thesame therapeutic benefits as the natural herb, which contains at least 100cannabinoids in addition to THC. Recent research conducted by GWPharmaceuticals in Great Britain has shown that Marinol is simply not aseffective for pain management as the whole plant; a balance of cannabi-noids, specifically CBC and CBD with THC, is what helps patients most. In
Americans for Safe Access
fact, Marinol is not labeled for pain, only appetite stimulation and nauseacontrol. But studies have found that many severely nauseated patients expe-rience difficulty in getting and keeping a pill down, a problem avoided withinhaled cannabis.
Clinical research on Marinol vs. cannabis has been limited by federal restric-tions, but a 2001 review of clinical trials conducted in the 70's and 80'sreports that "…the inhalation of THC appears to be more effective than theoral route."53 Additionally, patients frequently have difficulty getting theright dose with Marinol, while inhaled cannabis allows for easier titrationand avoids the negative side effects many report with Marinol. As the Houseof Lords observed, "Some users of both find cannabis itself more effective."
THE EXPERIENCE OF PATIENTS
Angel McClary Raich
I have been permanently disabled since September 1995. I am a mother oftwo teenage children. My children know more than anyone how medicalcannabis brought their mommy back to them. The hardest part of being dis-abled is watching the suffering in your children's eyes as they watch youendure such suffering with no end in sight.
In late 1997, my doctor felt cannabis would be an effective medication totreat my many complicated and complex medical conditions. I was in awheelchair from January 1996 to August 1999. Cannabis was responsible forgetting me out of my wheelchair and restoring mobility on the whole rightside of my body. For years I felt as if I was suffering in Hell. What I had toendure was unbelievable and indescribable torture.
I suffer greatly from severe chronic pain every single day. The prolonged painand suffering from my medical conditions significantly interferes with myquality of life. My treatment is complicated by the fact that I am violentlyallergic and have severe multiple chemical sensitivities to almost all phar-maceutical medicines. This interferes with the treatment of all of my med-ical conditions, and it means my suffering cannot be controlled by syn-thetic medications. This makes it extremely difficult for doctors to effec-tively help me combat my diseases. Without cannabis my life would be adeath sentence.54
In 1911, at the age of one, I contracted the polio virus. . The early onset ofpolio caused permanent damage in my legs, spine, and back, resulting in sig-nificant weakness and atrophy in my legs. As a result, I have never been ableto walk without the assistance of crutches and braces or a wheelchair.
Approximately 30 years ago, my condition began to deteriorate. I began to
suffer from increasing levels Angel Raich using a vaporizer in the hospital ofpain and weakness in my legs and back as well as severe osteoarthritis in myhands, arms, and joints. Over time, my deteriorating medical condition hasbeen exacerbated by my pain, leaving me increasingly immobilized.
By May, 1996, my physician [Dr. Arnold Leff, M.D.] had tried various prescrip-tion medications to relieve my pain, including: Tylenol #3, Ultram, Daypro,Tegretol, Soma, Valium, steroid injections into the trigger point, Dilantin,Duragesic, Zofran and Comapazine for the nausea caused by the opioid
pain relievers, and Doloboid andLodine as nonsteroids. Nothing
AMERICAN NURSES ASSOCIATION
seemed to work, and the painpersisted. I was growing increas-
In 2003 the American Nurses Association
ingly depressed by the inability of
passed a resolution that supports thosehealth care providers who recommend
anything to relieve my pain.
medicinal use, recognizes "the right of
During this period it was clear to
patients to have safe access to therapeu-
me, my caretaker, and my physi-
tic marijuana/cannabis," and calls for
cian that nothing was working to
more research and education, as well as arescheduling of marijuana for medical use.
combat my pain. My caretaker,Pat, had heard of the successsome people experience with the
medicinal use of marijuana for pain management. Sometime during theend of 1997, she obtained a sample for me. Although I had never used mar-ijuana in my previous eighty-seven years of life, I was willing to try anythingthat could alleviate even part of the pain.
The relief I experienced from medical marijuana was almost immediate. Iwas so pleased with the result that I wrote to Dr. Leff about my use of med-ical marijuana and we talked about the benefits of the medicine. Dr. Leffexamined me and noted that medical marijuana helped me experience lesschronic pain and nausea, leading him to recommended medical marijuanaas part of my daily pain care regimen.
I strongly feel that I should have the right to use anything that may relieve anyor some of my pain, and my last days should not be spent suffering. In 1998,around the time that I had to stop using the Duragesic patch, Dr. Leff pre-scribed 5 milligram tablets of Marinol, to be taken as needed, for pain man-agement. He explained that Marinol was like marijuana, which I was alreadyusing on occasion. Although Marinol provided me with some minor relief frommuscle spasms and bodily pains, its effect was slow and unpredictable. Attimes, however, I am stricken with severe spasms of pain, and medical marijua-na is the only medication that provides quick and effective relief. Medicalmarijuana also combats the nausea that accompanies many of the oral medica-tions I am prescribed, including anti-inflammatory medications such as Motrin.
Ever since trying medical marijuana, my life has drastically improved.
Although chronic pain, related to my post-polio syndrome will always
Americans for Safe Access
be a part of my life, medical marijuana had helped me manage thispain by providing fast and effective relief for my muscle spasms, acutepains, and arthritis.
Since I began using medical marijuana, my pain is no longer persistent ordebilitating. When I do suffer from pain, I am usually able to "get ahead ofit" by using medical marijuana and make it manageable.55
James Daniel Baehr
In 1994, I was diagnosed with inoperable prostate cancer. the cancer hadmetastasized to my spine, hips, and ribcage. The neuropathic back pain wasexcruciating, emanating from my spine to my hips and ribcage. I also experi-enced an overall loss of strength that substantially limited my ability towork. Employment in the transportation industry involves a considerableamount of carrying, lifting, and other manual labor that requires flexibilityand mobility. The performance of these requirements exacerbated the mag-nitude and amount of pain I experienced on a daily basis and depleted anyenergy that had not already been beaten down by the disease itself.
I began taking numerous medications to treat the cancer, the excruciatingpain that it caused, and the depression I felt as a result of my prognosis andthe profound restrictions on my life. My medications included a dailydosage of 7.5 mg of Lortab (a painkiller), .25 mg of Xanax (which combatsdepression and anxiety), 40 mg of Paxil (an anti-depressant), and 250 mg ofEulexin (which treats the cancer by reducing the testosterone emitted fromadrenal glands), and monthly shots of 7.5 mg of Lupron Depot (a testos-terone blocker/hormonal therapy). I suffered various side effects from thesemedications, including persistent exhaustion, general pain, a lack of mentalfocus, and overall body tenderness. In combination, these side effects werequite debilitating.
From September through December of 1995, I endured nine weeks of radia-tion. The treatment left me with continued back pain, intense nausea, lossof appetite, diverticulitis, sleep abnormalities, and digestive and intestinalcomplications. It also left me increasingly depressed.
In late 1994 or 1995, a physician at the Radiology Department at StanfordUniversity Hospital prescribed Marinol to alleviate my pain and nausea fromthe radiation. I tried the Marinol but did not respond well to it. Not only didMarinol make me feel drugged and not in control of my thoughts or body,but it failed to relieve my painful symptoms. In fact, Marinol just made mefeel sicker, upsetting my stomach, disrupting my mental acuity, and causingme to hallucinate. During this period, I was also taking 7.5 mg of Lortab, anopioid analgesic, several times a day and Ambien to help me sleep. Thesedrugs alleviated the pain somewhat, but also made me disoriented, consti-pated, and caused me to lose my short-term memory and fine motor skills.
Perhaps sensing that my hope was receding as my misery was increasing, anurse at Stanford Hospital suggested that medical marijuana could alleviatemy nausea, restore my appetite, and even help me manage my pain - allpotentially without the negative side effects I experienced with Marinol andother medications.
I decided to try a small amount of medical marijuana, and when I did Ifound that it provided significant relief from the side effects of the cancermedications and the radiation treatment. In addition, it helped reduce thepain I was experiencing from the cancer itself. This new combination oftherapies, which included medical marijuana, turned my health around.
Where before I had been doubled over with nausea, couldn't eat, or sleep, Iwas now not only able to handle my medications, but could sleep, eat andmanage my pain. I found that a small amount of medical marijuana takenin the evening enabled me to sleep through the entire night so that I nolonger needed to take Ambien.
Over time, the pain got progressively worse. In February 1997 I began totake morphine to help with the pain. The amount of morphine that I needto take to adequately control my pain leaves me utterly incapacitated, men-tally and physically. Medical marijuana helps me manage my pain, while lim-iting my dependence on more powerful narcotics.
When I smoke medical marijuana, I can achieve the same degree of painrelief with a much smaller amount of morphine and with far fewer and lessharsh side effects. The coupling of medical marijuana with my prescriptionanalgesics has been one of the most significant and successful aspects of mymedical treatment.56
THE EXPERIENCE OF DOCTORS
Dr. Harvey L. Rose
Both my research and my many years as a clinician have convinced me thatmarijuana can serve at least two important roles in safe and effective painmanagement. Ample anecdotal evidence and clinical observations, as wellas significant research findings, strongly indicate that marijuana, for what-ever reason, is often effective in relieving pain. This is true across a range ofpatient populations, including the elderly, the terminally ill seeking comfortin their final days, young adults stricken with life-threatening conditions,and cancer patients unable to tolerate the devastating effects of potentiallylife-saving therapies. Marijuana is also widely recognized as an antiemeticthat reduces the nausea and vomiting often induced by powerful opioidanalgesics prescribed for chronic, severe pain, as well as the nausea, vomit-ing and dizziness which often accompany severe and/or prolonged pain. Ihave had the benefit of consultations on this subject over many years with arange of treatment providers, including physicians, oncologists, pharmacolo-
Americans for Safe Access
gists, family practitioners, hospice workers, and pain specialists.
Specifically, I have found that cannabis can have an important opioid-spar-ing effect for pain patients. That is to say that patients who are prescribedhigh doses of opioid analgesics can significantly reduce their reliance onthese medications andimprove their daily function-ing by incorporating cannabis
NEW ENGLAND JOURNAL OF MEDICINE
into their pain care regimen.
"A federal policy that prohibits physicians
Marijuana not only has impor-
from alleviating suffering by prescribing
tant analgesic properties but
marijuana to seriously ill patients is mis-
it also is an effective and
guided, heavy-handed, and inhumane. It is
important adjuvant therapy
also hypocritical to forbid physicians toprescribe marijuana while permitting them
for patients suffering acute
to prescribe morphine and meperidine to
and/or chronic pain. No expe-
relieve extreme dyspnea and pain…there is
rienced and respected physi-
no risk of death from smoking marijuana.
cian will deny that for such
To demand evidence of therapeutic efficacy
patients opioid therapy is cen-
is equally hypocritical"
tral to palliative care. By thesame token, the same experi-
Jerome P. Kassirer, MD, editor
enced physicians will readily
N Engl J Med 336:366-367, 1997
acknowledge that opioidsoften induce nausea and vom-iting. For a number of pain patients, standard prescription antiemetics (e.g.,Compazine, Zofran and Reglan) simply do not substantially reduce their nau-sea. For many, those medications are substantially less effective, or producemore debilitating side effects, than marijuana.
Quite simply, marijuana can serve much the same function for pain patientsundergoing opiate therapy that it does for cancer patients undergoingchemotherapy: it suppresses the nausea and vomiting associated with treat-ment, and reduces the pain associated with prolonged nausea and retching,thereby increasing the chances that the patient will remain compliant withthe primary treatment. With both chemotherapy and long-term pain man-agement, failure to obtain and continue proper palliative and adjutant carecan have dire, even fatal, consequences.
Finally, it is important to note that in my clinical experience observingpatients who ingest cannabis for relief from pain and nausea and/or to stim-ulate appetite, I have witnessed no adverse complications. By contrast, manyof the first-line pharmaceuticals used to combat cancer, HIV/AIDS, and painassociated with these and other illnesses can induce a variety of iatrogeniceffects, including, in some instances, death. While patients may face seriouslegal implications related to their use of medical marijuana, as a physician Ihave yet to encounter a medical downside to their cannabinoid therapy.
[A]gainst the backdrop of a growing body of scientific research, the reports
of myriad pain patients, and the burgeoning clinical experience of physi-cians like myself, it is my considered opinion that cannabis can constitute anacceptable and sometimes necessary medicine to alleviate the immediatesuffering of certain patients.57
—Dr. Rose served as a medical officer in the Air Force before entering private prac-tice as a specialist on chronic pain. During his 40-year career, he has taught at the UCDavis School of Medicine and consulted with state legislative bodies in California,Idaho, Nevada, Washington and Oregon on pain management and the appropriaterole of regulatory agencies.
Richard I. Gracer, M.D.
For a small number of patients, even aggressive opiate therapies are notsufficient. Unless alternative pain treatments are found for such patients,they will continue to suffer. For those individuals, their daily lives are oftentortuous. As a physician, I am acutely aware of the disturbing connectionbetween intractable pain, overwhelming despair, and suicide.
I can state confidently, as a physician with an extensive practice and special-ized expertise in pain management, that marijuana can prove (and hasproven) medically useful to at least some chronic pain patients. Accordingly,I believe that physicians should be able to recommend and/or prescribe mar-ijuana to patients for whom it is medically appropriate. Absent that authori-ty, my ability to treat my patients and provide relief from horrific pain isundermined, as is the trust essential to therapeutic relationship.
—Dr. Gracer is Director of Orthopedic Medicine for ChiroView. He is a Fellow of theAmerican Academy of Family Physicians and a Diplomate of the American Academyof Pain Management.
Robert V. Brody, M.D.
As a physician responsible for the care and treatment of those who live inhorrible pain, I believe that these patients need, above all else, the broadestpossible range of therapeutic options and as full and accurate informationas possible regarding those options as they relate to the individual patient.
In recent years, I have noted that the public and the government havebecome increasingly aware of these needs, and one hopes that measureshave been taken to promote adequate pain care for the seriously ill andinjured. Several states, including California, have adopted laws and/orguidelines for the prescribing of controlled substances, which seem to per-mit physicians to treat pain patients without fear of sanction or interferencefrom state authorities.
Insofar as The Compassionate Use Act passed in 1996 expressly provides thatchronic pain is a condition for which physicians are authorized to recom-mend marijuana without threat or fear of punishment, the Act appears tobe an additional assurance for physicians like myself that we can rely upon a
Americans for Safe Access

full range of treatment modalities to care for patients in pain. The IOMReport provides still further support for doctors insofar as it recognizes thepotential medical benefits of marijuana. Marijuana has a place in any painphysician's armamentarium.58
—Dr. Brody is Chief of the Pain Consultation Clinic at San Francisco General Hospital.
He is a peer reviewer for the Western Journal of Medicine, Journal of GeneralInternal Medicine, Annals of Internal Medicine, and the Journal of Law, Medicineand Ethics.
THE HISTORY OF CANNABIS AS MEDICINE
The history of the medical use of cannabis dates back to 2700 B.C. in thepharmacopoeia of Shen Nung, one of the fathers of Chinese medicine. Inthe west, it has been recognized as a valued, therapeutic herb for centuries.
In 1823, Queen Victoria's personal physician, Sir Russell Reynolds, not onlyprescribed it to her for menstrual cramps but wrote in the first issue of TheLancet, "When pure and administered carefully, [it is] one of the of themost valuable medicines we possess."59
The American Medical Association opposed the first federal law againstcannabis with an article in its leading journal.60 Their representative, Dr.
William C. Woodward, testified to Congress that "The American MedicalAssociation knows of no evidence that marihuana is a dangerous drug,"and that any prohibition "loses sight of the fact that future investigationmay show that there are substantial medical uses for Cannabis." Cannabisremained part of the American pharmacopoeia until 1942 and is now avail-able by prescription in the Netherlands and Canada.
Federal Policy is Contradictory
Federal policy on medical cannabis is filled with contradictions. Cannabiswas widely prescribed until the turn of the century. Now cannabis is aSchedule I drug, classified as having no medicinal value and a high potentialfor abuse, yet its most psychoactive component, THC, is legallyavailable as Marinol and is classified as Schedule III. But the U.S.
federal government also grows and provides cannabis for a smallnumber of patients today.
In 1976 the federal government created the Investigational NewDrug (IND) compassionate access research program to allowpatients to receive medical cannabis from the government. Theapplication process was extremely complicated, and few physi-cians became involved. In the first twelve years the government acceptedabout a half dozen patients. The federal government approved the distribu-tion of up to nine pounds of cannabis a year to these patients, all of whomreport being substantially helped by it.
In 1989 the FDA was deluged with new applications from people with AIDS,and 34 patients were approved within a year. In June 1991, the PublicHealth Service announced that the program would be suspended because itundercut the administration's opposition to the use of illegal drugs. Theprogram was discontinued in March 1992 and the remaining patients hadto sue the federal government on the basis of "medical necessity" to retainaccess to their medicine. Today, a few surviving patients still receive medicalcannabis from the federal government, grown under a doctor's supervisionat the University of Mississippi and paid for by federal tax dollars.
Despite this successful medical program and centuries of documented safeuse, cannabis is still classified in America as a Schedule I substance.
Healthcare advocates have tried to resolve this contradiction through legaland administrative channels. In 1972, a petition was submitted to resched-ule cannabis so that it could be prescribed to patients. The DEA stalledhearings for 16 years, but in 1988 their chief administrative law judge,Francis L. Young, ruled that, "Marijuana, in its natural form, is one of thesafest therapeutically active substances known. It would be unreasonable,arbitrary and capricious for the DEA to continue to stand between thosesufferers and the benefits of this substance."
The DEA refused to implement this ruling based on a procedural technicali-ty and continues to classify cannabis as a substance with no medical use.
Widespread public support; state laws passed
Public opinion is clearly in favor of ending the prohibition of medicalcannabis and has been for some time. A CNN/Time poll in November 2002found that 80% of Americans support medical cannabis. The AARP, thenational association whose 35 million members are over the age of fifty,released a national poll in December 2004 showing that nearly two-thirds ofolder Americans support legal access to medical marijuana. Support in theWest, where most states that allow legal access are located, was strongest,at 82%, but at least 2 out of 3 everywhere agreed that "adults should beallowed to legally use marijuana for medical purposes if a physician recom-mends it."
The refusal of the federal government to act on this support has meant thatpatients have had to turn to the states for action. Since 1996, 15 states haveremoved criminal penalties for their citizens who use cannabis on the adviceof a physcian. Voters have passed medical cannabis ballot initiatives in 10states plus the District of Columbia, while the legislatures in Hawaii,Maryland, New Jersey, New Mexico, Rhode Island, and Vermont and haveenacted similar bills. Approximately one third of the U.S. population residesin a state that permits medical use, and medical cannabis legislation is intro-duced in more states every year.
Americans for Safe Access
Currently, laws that effectively remove state-level criminal penalties forgrowing and/or possessing medical cannabis are in place in Alaska, Arizona,California, Colorado, Hawaii, Maine, Montana, Nevada, New Jersey, NewMexico, Oregon, Rhode Island, Vermont, Washington, and the District ofColumbia. Maryland has reduced the criminal penalty for medical use to amaximum $100 fine. Thirty-six states have symbolic medical cannabis laws(laws that support medical cannabis but do not provide patients with legalprotection under state law).
2005 U.S. Supreme Court ruling
In June 2005, the U.S. Supreme Court overturned a decision by a U.S.
appeals court (Raich v. Ashcroft) that had exempted medical marijuana fromfederal prohibition. The 2005 decision, now called Gonzales v. Raich, ruledthat federal officials may prosecute medical marijuana patients for possess-ing, consuming, and cultivating medical cannabis. But courts have foundthat ruling does not affect individual states' medical marijuana programs,and only applies to prosecution in federal, not state, court.
Petitions for legal prescriptions pending
The federal Department of Health and Human Services (HHS) and the FDAare currently reviewing two legal petitions with broad implications for med-ical marijuana. The first, brought by ASA under the Data Quality Act, saysHHS must correct its statements that there is no medical use for marijuanato reflect the many studies which have found it helpful for many conditions.
Acknowledging legitimate medical use would then force the agency to con-sider allowing the prescribing of marijuana as they do other drugs, basedon its relative safety. A separate petition, of which ASA is a co-signer, asksthe Drug Enforcement Administration for a full, formal re-evaluation ofmarijuana's medical benefits, based on hundreds of recent medical researchstudies and two thousand years of documented human use.
Legal Citations1. See "The Administration's Response to the Passage of California Proposition 215 and
Arizona Proposition 200" (Dec. 30, 1996).
2. See Conant v. McCaffrey, 172 F.R.D. 681 (N.D. Cal. 1997).
3. See id.; Conant v. McCaffrey, 2000 WL 1281174 (N.D. Cal. 2000); Conant v. Walters, 309
F.3d 629 (9th Cir. 2002).
4. 309 F.3d 629 (9th Cir. 2002).
5. Id. at 634-36.
6. Criminal liability for aiding and abetting requires proof that the defendant "insome sort
associate[d] himself with the venture, that he participate[d] in it as something that hewishe[d] to bring about, that he [sought] by his action to make it succeed."Conant v.
McCaffrey, 172 F.R.D. 681, 700 (N.D. Cal. 1997) (quotation omitted). A conspiracy toobtain cannabis requires an agreement between two or more persons to do this, withboth persons knowing this illegal objective and intending to help accomplish it. Id. at700-01.
7. 309 F.3d at 634 & 636.
8. Conant v. McCaffrey, 2000 WL 1281174, at *16 (N.D. Cal. 2000).
9. 309 F.3d at 634.
10. See id. at 635; Conant v. McCaffrey, 172 F.R.D. 681, 700-01 (N.D. Cal. 1997).
Research Citations
11.Abrams DI et al (2003). Short-Term Effects of Cannabinoids in Patients with HIV-1
Infection: A Randomized, Placebo-Controlled Clinical Trial. Ann Intern Med. Aug19;139(4):258-66.5.
12. Aggrawal S et al. 2009. Medicinal use of cannabis in the United States: histori-
cal perspectives, current trends, and future directions. J Opioid Manag. May-Jun;5(3):153-68.
13.Russo EB. (2008) Cannabinoids in the management of difficult to treat pain. Therap
and Clincial Risk Manag 4(1) 245-259.
14.Barnes MP (2006). Sativex: clinical efficacy and tolerability in the treatment of symp-
toms of multiple sclerosis and neuropathic pain. Expert Opin Pharmacother, 7:607-15.
15.Perez J (2006) Combined cannabinoid therapy via an oral mucosal spray. Drugs
Today (Barc.), 42:495-501.
16. Selvarajah D et al (2010). Randomized placebo-controlled double-blind clinical trial
of cannabis-based medicinal product (Sativex) in painful diabetic neuropathy.
Diabetes Care. 33(1):128-30.
17. Russo 2008. Op cit.
18.Dixon WE (1899). The pharmacology of Cannabis indica. BMJ, ii: 1354-1357. 19.O'Shaughnessy WB (1838). On the preparations of the Indian hemp, or gunjah
(Cannabis indica); their effects on the animal system in health, and their utility inthe treatment of tetanus and other convulsive diseases. Transactions of the Medicaland Physical Society of Bengal 18; 40: 71-102, 421-61.
20.Reynolds JR (1890) Therapeutical uses and toxic effects of Cannabis indica. Lancet, i:
21.Noyes R et al (1975). The analgesic properties of delta-9-tetrahydrocannabinol and
codeine. Clinical Pharmacology and Therapeutics, 18: 84-89.
22.Noyes R, Baram D (1974). Cannabis analgesia. Compr. Psychiatry 15: 531. 23.Petro D (1980). Marihuana as a therapeutic agent for muscle spasm and spasticity.
Psychosomatics 21 81-85.
24.El-Mallakh R (1987). Marijuana and migraine. Headache, 27 442-443. 25.Holdcroft A et al (1997). Pain relief with oral cannabinoids in familial
Mediterranean fever. Anaesthesia, 5 483-486.
26.Hall W et al (1994). The Health and Psychological Consequences of Cannabis Use.
Canberra, Australian Government Publishing Service.
27.Society for Neuroscience Press Conference, October 26, 1997.
28. Joy J et al (1999). Marijuana and Medicine: Assessing the Science Base. Washington
D.C. National Academy Press.
29.Martin-Sanchez E, Toshiaki A., et al (2009) Systematic Review and Meta-analysis of
Cannabis Treatment for Chronic Pain. Pain Medicine.
30. Ware M, Wang W, Shapiro S, et al (2007). Smoked cannabis for chronic neuropathic
pain: results of a pilot study. 17th Annual Symposium on the Cannabinoids. Saint-Sauveur, Quebec, Canada: International Cannabinoid research Society p31.
31.Growing L et al (1998). Therapeutic use of cannabis: clarifying the debate. Drug and
Alcohol Review. 17 445-452.
32. Rahn EJ Hohmann AG. 2009. Cannabinoids as pharmacotherapies for neuropathic
pain: from the bench to the bedside. Neurotherapeutics. Oct;6(4):713-37.
33.Abrams DI, Jay CA, Shade SB et al (2007). Cannabis in painful HIV-associated senseo-
ry neuropathy: a randomized placebo-controlled trial. Neurology, 68:515-21.
34.Cookson C (2001). High Hopes for Cannabis to Relieve Pain. British Association
Americans for Safe Access
Science Festival in Glasgow, Financial Times, September 4, at National News pg. 4.
35.Ibid. Russo 2008.
36.Gomez MA, Saenz MT et al (1999). Study of the topical anti-inflammaotry activity of
achillea ageratum on chronic and acute inflammation models. Z Naturforsch , 54:937-41.
37.Barrett ML, Scutt AM et al (1988) Cannaflavin A and B, prenylated flavones from
Cannabis Sativa L. Expermentia, 42:452-3.
38.Gertsch J. (2008) Anti-inflammatory Xannabinoids in Diet. Communicative &
Intergrative Biology 2008 vol.1 issue 1.
39.Karst M et al (2003). Analgesic Effect of the Synthetic Cannabinoid CT-3 on Chronic
Neuropathic Pain A Randomized Controlled Trial. JAMA. 290:1757-1762.
40.Richardson J et al (1998). Cannabinoids Reduce Hyperalgesia and Inflammation via
Interaction with Peripheral CB1 Receptors. Pain. 75(1): 111-119.
41.Meng I et al (1998). An analgesic circuit activated by cannabinoids. Nature 395 381-383.
42.Klarreich E (2001). Cannabis spray blunts pain: Early trials suggest cannabis spritz may
give relief to chronic pain sufferers. British Association for the Advancement of Science.
43.Callahan R (1998). "How Does Marijuana Kill Pain?" Associated Press, October 4.
44.Welch SP, Eads M (1999). Synergistic interactions of endogenous opioids and cannabi-
noid systems. Brain Res. Nov. 27;848 (1-2):183-90.
45.Maurer et al. (1990). Delta-9-tetrahydrocannabinol Shows Antispastic and Analgesic
Effects in a Single Case Double-Blind Trial. European Archives of Psychiatry and ClinicalNeuroscience 240:1-4
46.Holdcroft, A., op cit. 47.Martin WJ (1999). Basic Mechanisms of Cannabinoid-Induced Analgesia. International
Association for the Study of Pain Newsletter, Summer. p. 89.
48.House of Lords Select Committee on Science and Technology, "Ninth Report" (1998).
London: United Kingdom. Section 5.26.
49. Grinspoon L et al. (1995). Marihuana as medicine. A plea for reconsideration. JAMA.
50.Hazekamp A et al (2006). Evaluation of a vaporizing device (Volcano(R)) for the pul-
monary administration of tetrahydrocannabinol. J Pharm Sci 95 (6) Apr 24: 1308-1317.
51.Sidney S et al (1997). Marijuana Use and Cancer Incidence. Cancer Causes and Control;
52.Tashkin D (2006). Marijuana Use and Lung Cancer: Results of a Case-Control Study.
American Thoracic Society International Conference. May 23, 2006.
53.Musty R, Rossi R (2001). Effects of smoked cannabis and oral delta-9-tetrahydrocannabi-
nol on nausea and emesis after cancer chemotherapy: a review of state clinical trials.
Journal of Cannabis Therapeutics. 1: 29-56.
54.Statement of Angel McClary Raich. From angeljustice.com, website for her suit, Angel
McClary Raich, et. al. vs. John Ashcroft, et. al. United States District Court of the NorthDistrict of California. No. C 02-4872. United States Court of Appeals for the NinthDistrict. No. 03-15481. Retrieved fromhttp://angeljustice.com/article.php?list=type&type=6.
55.Declaration of Dorothy Gibbs. County of Santa Cruz et. al. vs. Ashcroft et. al. United
States Court of Appeals for the Ninth District. Retrieved from http://www.san-tacruzvsashcroft.com/pleadings_gibbs.htm.
56.Declaration of James Daniel Baehr. Ibid. Retrieved from http://www.san-
57.Declaration of Robert Brody, M.D. Ibid. Retrieved from http://www.san-
58.Declaration of Harvey L. Rose, M.D. Ibid. Retrieved from http://www.san-
59.Lancet 1; 1823. 60.108 J.A.M.A. 1543-44; 1937.
DEA CHIEF ADMINISTRATIVE LAW JUDGE
Marijuana, in its natural form, is one of the safest therapeu-tically active substances known. It would be unreasonable,arbitrary and capricious for the DEA to continue to stand between those sufferers and the benefits of this substance.
The Honorable Francis L. Young,Ruling on DEA rescheduling hearings, 1988
ADDITIONAL RESOURCES
Americans for Safe Access maintains a website with additionalresources for doctors and patients. There you will find thelatest information on legal and legislative developments, newmedical research, and what you can do to help protect therights of patients and doctors.
With more than 45,000 active members and chapters and affil-iates in all 50 states, ASA is the largest national member-based organization of patients, medical professionals, scientists, andconcerned citizens promoting safe and legal access to cannabisfor therapeutic uses and research.
1322 Webster Street, Suite 402, Oakland, California 94612
rev. Feb 2011
Source: http://www.medicalmarijuanaoftucson.com/wp-content/uploads/2015/12/pain_brochure.pdf
European Journal of Echocardiography (2009) 10, 194–212doi:10.1093/ejechocard/jep005 EAE RECOMMENDATIONS Contrast echocardiography: evidence-basedrecommendations by European Associationof Echocardiography Roxy Senior1*, Harald Becher2, Mark Monaghan3, Luciano Agati4, Jose Zamorano5,Jean Louis Vanoverschelde6, and Petros Nihoyannopoulos7 1Department of Cardiology, Northwick Park Hospital, Imperial College, London, Harrow HA1 3UJ, UK; 2John RadcliffeHospital, Oxford, UK; 3King's College Hospital, London, UK; 4La Sapienza University, Rome, Italy; 5Hospital Clı´nico San Carlos,Madrid, Spain; 6Cliniques Universitaires St-Luc, Universite
Fichas Internacionales de Seguridad Química BENZO [a] ANTRACENO ICSC: 0385 BENZO a ANTRACENO 1,2-Benzoantraceno 2,3-Benzofenantreno Masa molecular: 228.3 Nº CAS 56-55-3 Nº RTECS CV9275000 Nº ICSC 0385 Nº NU 2811 PELIGROS/ SINTOMAS PRIMEROS AUXILIOS/ PELIGRO/ PREVENCION










